Adsorption of Serum Albumin onto Octacalcium Phosphate in Supersaturated Solutions Regarding Calcium Phosphate Phases
- PMID: 31340468
- PMCID: PMC6678651
- DOI: 10.3390/ma12142333
Adsorption of Serum Albumin onto Octacalcium Phosphate in Supersaturated Solutions Regarding Calcium Phosphate Phases
Abstract
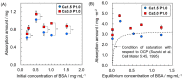
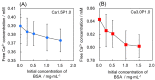
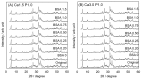
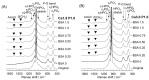
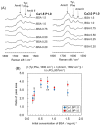
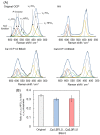
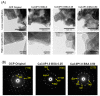
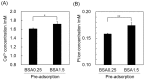
Octacalcium phosphate (OCP) has been shown to enhance new bone formation, coupled with its own biodegradation, through osteoblasts and osteoclast-like cell activities concomitant with de novo hydroxyapatite (HA) formation and serum protein accumulation on its surface. However, the nature of the chemical environment surrounding OCP and how it affects its metabolism and regulates protein accumulation is unknown. The present study examined how the degree of supersaturation (DS) affects the bovine serum albumin (BSA) adsorption onto OCP in 150 mM Tris-HCl buffer at 37 °C and pH 7.4, by changing the Ca2+ ion concentration. The amount of BSA adsorbed onto OCP increased as the DS increased. In addition, the amount of newly formed calcium phosphate, which could be OCP, was increased, not only by increases in DS, but also at lower equilibrium concentrations of BSA. The increased adsorption capacity of BSA was likely related to the formation of calcium phosphate on the adsorbed OCP. Together the results suggested that the formation of new calcium phosphate crystals is dependent on both the DS value and the adsorbate protein concentration, which may control serum protein accumulation on the OCP surface in vivo.
Keywords: bovine serum albumin; degree of supersaturation; octacalcium phosphate; protein adsorption.
Conflict of interest statement
The authors declare no conflict of interest but the content of the present study was used for a patent application.
Figures

References
-
- Brown W.E., Smith J.P., Frazier A.W., Lehr J.R. Crystallographic and chemical relations between octacalcium phosphate and hydroxyapatite. Nature. 1962;196:1050–1055. doi: 10.1038/1961050a0. - DOI
-
- Brown W.E., Mathew M., Tung M.S. Crystal chemistry of octacalcium phosphate. Prog. Cryst. Growth Charact. 1981;4:59–87. doi: 10.1016/0146-3535(81)90048-4. - DOI
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

