Phase 1 study of the selective BTK inhibitor zanubrutinib in B-cell malignancies and safety and efficacy evaluation in CLL
- PMID: 31340982
- PMCID: PMC6742923
- DOI: 10.1182/blood.2019001160
Phase 1 study of the selective BTK inhibitor zanubrutinib in B-cell malignancies and safety and efficacy evaluation in CLL
Abstract
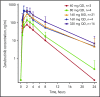
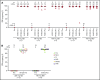
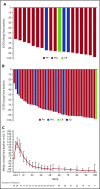
Zanubrutinib is a potent and highly selective inhibitor of Bruton tyrosine kinase (BTK). In this first-in-human, open-label, multicenter, phase 1 study, patients in part 1 (3 + 3 dose escalation) had relapsed/refractory B-cell malignancies and received zanubrutinib 40, 80, 160, or 320 mg once daily or 160 mg twice daily. Part 2 (expansion) consisted of disease-specific cohorts, including treatment-naive or relapsed/refractory chronic lymphocytic leukemia/small lymphocytic lymphoma (CLL/SLL). The primary end points were safety and tolerability, and definition of the maximum tolerated dose (part 1). Additional end points included pharmacokinetics/pharmacodynamics and preliminary efficacy. Reported herein are results from 144 patients enrolled in the dose-finding and CLL/SLL cohorts. No dose-limiting toxicities occurred in dose escalation. Median BTK occupancy in peripheral blood mononuclear cells was >95% at all doses. Sustained complete (>95%) BTK occupancy in lymph node biopsy specimens was more frequent with 160 mg twice daily than 320 mg once daily (89% vs 50%; P = .0342). Consequently, 160 mg twice daily was selected for further investigation. With median follow-up of 13.7 months (range, 0.4-30.5 months), 89 CLL/SLL patients (94.7%) remain on study. Most toxicities were grade 1/2; neutropenia was the only grade 3/4 toxicity observed in >2 patients. One patient experienced a grade 3 subcutaneous hemorrhage. Among 78 efficacy-evaluable CLL/SLL patients, the overall response rate was 96.2% (95% confidence interval, 89.2-99.2). Estimated progression-free survival at 12 months was 100%. Zanubrutinib demonstrated encouraging activity in CLL/SLL patients, with a low incidence of major toxicities. This trial was registered at www.clinicaltrials.gov as #NCT02343120.
© 2019 by The American Society of Hematology.
Conflict of interest statement
Conflict-of-interest disclosure: C.S.T. reports research funding, personal fees, and other from BeiGene during the conduct of the study, and personal fees from Janssen and Pharmacyclics outside of the submitted work. G.C. reports research funding from BeiGene during the conduct of the study, served on an advisory board for AbbVie Pty Ltd, and received travel grants from Amgen Australia and Takeda Australia outside of the submitted work. P.M. reports personal fees and nonfinancial support from Novartis, Roche, Janssen, Amgen, and Celgene outside of the submitted work. J.M. reports personal fees from Gilead/Kite Pharma, Pharmacyclics/Janssen, Bayer, Alexion, Pfizer, Juno/Celgene, Bristol-Myers Squibb, Genentech, and Kyowa outside of the submitted work. S.O. reports research funding from BeiGene during the conduct of the study. J.F.S. reports grants, personal fees, and other from AbbVie and Janssen, and personal fees and other from Acerta and Sunesis outside of the submitted work. D.S. reports grants from BeiGene during the conduct of the study; grants from Amgen, Acerta, Pharmacyclics, Millennium, Sanofi, and BeiGene; and grants and personal fees from AbbVie, Janssen, Roche, Celgene, and MSD outside of the submitted work. A.W.R. reports research funding from BeiGene during the conduct of the study; reports grants from AbbVie and Janssen; reports other from Genentech outside of the submitted work; and is an employee of the Walter and Eliza Hall Institute, which receives milestone and royalty payments relating to venetoclax. R.E., Z.T., L.H., J.H., and W.N. are employees of BeiGene USA. L.W. is an employee of BeiGene Beijing. Y.Y. is an employee of BeiGene Shanghai. The remaining authors declare no competing financial interests.
Figures

References
-
- Bernard S, Danglade D, Gardano L, et al. Inhibitors of BCR signalling interrupt the survival signal mediated by the micro-environment in mantle cell lymphoma. Int J Cancer. 2015;136(12):2761-2774. - PubMed
-
- de Rooij MF, Kuil A, Geest CR, et al. The clinically active BTK inhibitor PCI-32765 targets B-cell receptor- and chemokine-controlled adhesion and migration in chronic lymphocytic leukemia. Blood. 2012;119(11):2590-2594. - PubMed
Publication types
MeSH terms
Substances
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

