Functional importance of the oligomer formation of the cyanobacterial H+ pump Gloeobacter rhodopsin
- PMID: 31341208
- PMCID: PMC6656774
- DOI: 10.1038/s41598-019-47178-5
Functional importance of the oligomer formation of the cyanobacterial H+ pump Gloeobacter rhodopsin
Abstract
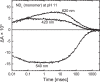
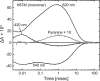
Many microbial rhodopsins self-oligomerize, but the functional consequences of oligomerization have not been well clarified. We examined the effects of oligomerization of a H+ pump, Gloeobacter rhodopsin (GR), by using nanodisc containing trimeric and monomeric GR. The monomerization did not appear to affect the unphotolyzed GR. However, we found a significant impact on the photoreaction: The monomeric GR showed faint M intermediate formation and negligible H+ transfer reactions. These changes reflected the elevated pKa of the Asp121 residue, whose deprotonation is a prerequisite for the functional photoreaction. Here, we focused on His87, which is a neighboring residue of Asp121 and conserved among eubacterial H+ pumps but replaced by Met in an archaeal H+ pump. We found that the H87M mutation removes the "monomerization effects": Even in the monomeric state, H87M contained the deprotonated Asp121 and showed both M formation and distinct H+ transfer reactions. Thus, for wild-type GR, monomerization probably strengthens the Asp121-His87 interaction and thereby elevates the pKa of Asp121 residue. This strong interaction might occur due to the loosened protein structure and/or the disruption of the interprotomer interaction of His87. Thus, the trimeric assembly of GR enables light-induced H+ transfer reactions through adjusting the positions of key residues.
Conflict of interest statement
The authors declare no competing interests.
Figures






References
Publication types
MeSH terms
Substances
Supplementary concepts
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

