A Cell-Based Renilla Luminescence Reporter Plasmid Assay for High-Throughput Screening to Identify Novel FDA-Approved Drug Inhibitors of HPV-16 Infection
- PMID: 31361520
- PMCID: PMC6925341
- DOI: 10.1177/2472555219860771
A Cell-Based Renilla Luminescence Reporter Plasmid Assay for High-Throughput Screening to Identify Novel FDA-Approved Drug Inhibitors of HPV-16 Infection
Abstract
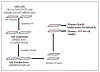
Like cervical cancer, anal cancer is caused by human papillomavirus (HPV). HPV is the most common sexually transmitted agent and is found in the anal canal of almost all HIV-positive men who have sex with men (MSM). Rates of HPV anal cancer are disproportionately higher in this population. Although the nanovalent HPV vaccine is efficacious in protecting against oncogenic HPV types, a substantial proportion of MSM remains unvaccinated and anal HPV infection continues to be an important public health burden. Therefore, it is important to identify strategies to prevent HPV infection. We report on two promising and interlinked strategies: (1) the development of a cell-based Renilla luminescence reporter assay using HPV-16 pseudovirions that encapsidate SV40-driven Renilla luminescence reporter expression plasmid and (2) use of this assay for high-throughput screening (HTS) of FDA- and internationally approved drugs to identify those that could be repurposed to prevent HPV infection. We conducted a screen of 1906 drugs. The assay was valid with a Z' of 0.67 ± 0.04, percent coefficient of variance of 10.0, and signal-to-background noise window of 424.0 ± 8.0. Five drugs were chosen for further analyses based on selection parameters of ≥77.0% infection of HPV-16 pseudovirion-driven Renilla expression with <20.0% cytotoxicity. Of these, the antifungal pentamidine and a gamma-amino butyric acid receptor agonist securinine exhibited ≥90.0% infection with <10.0% cytotoxicity. This luminescent cell-based reporter expression plasmid assay for HTS is a valid method to identify FDA- and internationally approved drugs with the potential to be repurposed into prevention modalities for HPV infection.
Keywords: HPV-16; high-throughput screening; papillomavirus; pseudovirus; repurposed.
Conflict of interest statement
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Figures
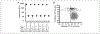
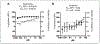
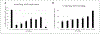
Similar articles
-
Identification of several high-risk HPV inhibitors and drug targets with a novel high-throughput screening assay.PLoS Pathog. 2017 Feb 9;13(2):e1006168. doi: 10.1371/journal.ppat.1006168. eCollection 2017 Feb. PLoS Pathog. 2017. PMID: 28182794 Free PMC article.
-
High-throughput drug screening using the Ebola virus transcription- and replication-competent virus-like particle system.Antiviral Res. 2018 Oct;158:226-237. doi: 10.1016/j.antiviral.2018.08.013. Epub 2018 Aug 24. Antiviral Res. 2018. PMID: 30149038
-
Prevalence of Anal Human Papillomavirus Infection and Risk Factors among HIV-positive Patients in Tokyo, Japan.PLoS One. 2015 Sep 14;10(9):e0137434. doi: 10.1371/journal.pone.0137434. eCollection 2015. PLoS One. 2015. PMID: 26368294 Free PMC article. Clinical Trial.
-
Human Papillomavirus and Genital Warts: A Review of the Evidence for the 2015 Centers for Disease Control and Prevention Sexually Transmitted Diseases Treatment Guidelines.Clin Infect Dis. 2015 Dec 15;61 Suppl 8:S849-55. doi: 10.1093/cid/civ813. Clin Infect Dis. 2015. PMID: 26602622 Review.
-
Human Papillomavirus Biology, Pathogenesis, and Potential for Drug Discovery: A Literature Review for HIV Nurse Clinical Scientists.J Assoc Nurses AIDS Care. 2015 Nov-Dec;26(6):693-702. doi: 10.1016/j.jana.2015.07.001. Epub 2015 Jul 8. J Assoc Nurses AIDS Care. 2015. PMID: 26277046 Free PMC article. Review.
Cited by
-
Nanotechnological approaches for pentamidine delivery.Drug Deliv Transl Res. 2022 Aug;12(8):1911-1927. doi: 10.1007/s13346-022-01127-4. Epub 2022 Feb 25. Drug Deliv Transl Res. 2022. PMID: 35217992 Free PMC article. Review.
-
Development of a Cell-Based Reporter Potency Assay for Live Virus Vaccines.Vaccines (Basel). 2024 Jul 13;12(7):769. doi: 10.3390/vaccines12070769. Vaccines (Basel). 2024. PMID: 39066407 Free PMC article.
-
Small Molecule Inhibitors of Human Papillomavirus: A Review of Research from 1997 to 2021.Curr Med Chem. 2024;31(33):5308-5350. doi: 10.2174/0929867331666230713165407. Curr Med Chem. 2024. PMID: 37448363 Review.
References
-
- Gervaz P; Hirschel B; Morel P Molecular Biology of Squamous Cell Carcinoma of the Anus. Br. J. Surg. 2006, 93, 531–538. - PubMed
-
- Bernardi M-P; Ngan SY; Michael M; et al. Molecular Biology of Anal Squamous Cell Carcinoma: Implications for Future Research and Clinical Intervention. Lancet Oncol. 2015, 16, e611–e621. - PubMed
-
- National Institutes of Health, National Cancer Institute. HPV and Cancer. https://www.cancer.gov/about-cancer/causes-prevention/risk/infectious-ag... (accessed June 30, 2019).

