WDR76 degrades RAS and suppresses cancer stem cell activation in colorectal cancer
- PMID: 31362761
- PMCID: PMC6668196
- DOI: 10.1186/s12964-019-0403-x
WDR76 degrades RAS and suppresses cancer stem cell activation in colorectal cancer
Abstract
Background: Stabilization of RAS is a key event for the hyper-activation of Wnt/β-catenin signaling and activation of cancer stem cell (CSC) in colorectal cancer (CRC). WD Repeat protein 76 (WDR76) mediates the polyubiquitination-dependent degradation of RAS in hepatocellular carcinoma (HCC). We investigated whether WDR76 destabilizes RAS and acts as a tumor suppressor inhibiting CSC activation in CRC.
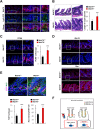
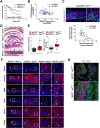
Methods: We generated mice with deletion of Wdr76 (Wdr76-/-) and crosses of Wdr76-/- with ApcMin/+ (Wdr76-/-; ApcMin/+) and compared them with wildtype mice (Wdr76+/+) and ApcMin/+ mice (Wdr76+/+; ApcMin/+), respectively. Intestinal crypt lengthening, tumorigenesis and CSC activation were analyzed by histology, immunohistochemistry, and immunoblotting. CRC cell line was engineered to stably express or knockdown WDR76 or control vector and was analyzed after spheroid culture.
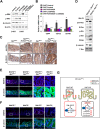
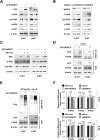
Results: Wdr76-/- mice, with increased Ras level, displayed crypt elongation and hyper-proliferation. Wdr76-/-; ApcMin/+ mice developed more tumors with bigger sizes than ApcMin/+ mice and their tumors showed increased proliferation and CSC activation with elevated RAS and β-catenin levels. In CRC cells, overexpression or knockdown of WDR76 decreased or increased the numbers and sizes of CRC spheroids with inhibition or activation of CSC markers, respectively. In human CRC, lower level of WDR76 was associated with poor patient survival.
Conclusions: In analyses of mice with deletion of Wdr76 and CRC spheroids, we found that RAS stability plays important roles in tumorigenesis by affecting proliferation and CSC activation. Our results suggest that destabilization of RAS by WDR76 is a potential strategy for targeting malignant CRC involving CSC activation.
Keywords: Cancer stem cell; Colorectal Cancer; RAS stability; Tumor suppressor; WDR76.
Conflict of interest statement
The authors declare that they have no competing interests.
Figures

References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases

