Efficacy and safety of plerixafor for hematopoietic stem cell mobilization for autologous transplantation in patients with non-Hodgkin lymphoma and multiple myeloma: A systematic review and meta-analysis
- PMID: 31363366
- PMCID: PMC6614714
- DOI: 10.3892/etm.2019.7691
Efficacy and safety of plerixafor for hematopoietic stem cell mobilization for autologous transplantation in patients with non-Hodgkin lymphoma and multiple myeloma: A systematic review and meta-analysis
Abstract
Plerixafor in combination granulocyte-colony stimulating factor (G-CSF) has been used for the mobilization of hematopoietic stem cells (HSCs) to the peripheral blood for collection and subsequent autologous transplantation in patients with non-Hodgkin lymphoma (NHL) and multiple myeloma (MM). The aim of this study was to systematically search the published literature and analyze evidence on the efficacy of additional plerixafor for successful HSC mobilization in patients with NHL and MM, and to evaluate the safety of the drug. The PubMed, Scopus, Cochrane Central Register of Controlled Trials (CENTRAL) and Google scholar databases were searched electronically for studies published in the English language up to March, 2019. Five studies (3 on NHL and 2 on MM) were included in this review article. The meta-analysis of data of 364 patients in the treatment group and 368 patients in the control group, indicated that the mobilization of ≥5/6×106 CD34+ cells/kg in 4 or less apheresis days was superior with plerixafor + G-CSF than with G-CSF alone (RR=2.59, 95% CI: 1.40 to 4.81; P<0.0001). Similarly, a greater proportion of patients in the treatment group exhibited the mobilization of ≥2×106 CD34+ cells/kg in 4 or less apheresis days (RR=1.46, 95% CI: 1.01 to 2.12; P=0.04). The addition of plerixafor significantly increased the total collection of CD34+ cells (random: MD=4.21; 95% CI: 2.85 to 5.57; P<0.00001). Meta-analysis indicated no significant increase in adverse events with the addition of plerixafor for HSC mobilization (RR=1.03, 95% CI: 0.99 to 1.06; P=0.16). On the whole, the findings of this study indicate that the addition of plerixafor to G-CSF leads to an increased HSC collection in a shorter period of time with no concomitant increase in adverse events. Further randomized controlled trials with a larger sample size evaluating short term efficacy, as well as long term survival would help to further strengthen the evidence on this subject.
Keywords: hematopoietic stem cell; lymphoma; multiple myeloma; plerixafor.
Figures


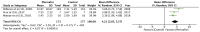
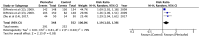
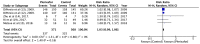
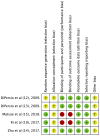
References
-
- Duong HK, Savani BN, Copelan E, Devine S, Costa LJ, Wingard JR, Shaughnessy P, Majhail N, Perales MA, Cutler CS, et al. Peripheral blood progenitor cell mobilization for autologous and allogeneic hematopoietic cell transplantation: Guidelines from the American Society for Blood and Marrow Transplantation. Biol Blood Marrow Transplant. 2014;20:1262–1273. doi: 10.1016/j.bbmt.2014.05.003. - DOI - PubMed
-
- Hosing C, Saliba RM, Ahlawat S, Körbling M, Kebriaei P, Alousi A, De Lima M, Okoroji JG, McMannis J, Qazilbash M, et al. Poor hematopoietic stem cell mobilizers: A single institution study of incidence and risk factors in patients with recurrent or relapsed lymphoma. Am J Hematol. 2009;84:335–337. doi: 10.1002/ajh.21400. - DOI - PMC - PubMed
-
- Pusic I, Jiang SY, Landua S, Uy GL, Rettig MP, Cashen AF, Westervelt P, Vij R, Abboud CN, Stockerl-Goldstein KE, et al. Impact of mobilization and remobilization strategies on achieving sufficient stem cell yields for autologous transplantation. Biol Blood Marrow Transplant. 2008;14:1045–1056. doi: 10.1016/j.bbmt.2008.07.004. - DOI - PubMed
LinkOut - more resources
Full Text Sources
