CONSORT 2010 statement: extension to randomised crossover trials
- PMID: 31366597
- PMCID: PMC6667942
- DOI: 10.1136/bmj.l4378
CONSORT 2010 statement: extension to randomised crossover trials
Abstract
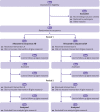
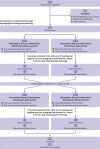
Evidence shows the quality of reporting of randomised controlled trials is not optimal. The lack of transparent reporting impedes readers from judging the reliability and validity of trial findings, prevents researchers from extracting information for systematic reviews, and results in research waste. The Consolidated Standards of Reporting Trials (CONSORT) statement was developed to improve the reporting of randomised controlled trials. The primary focus of the statement was on parallel group trials with two treatment groups. Crossover trials are a particular type of trial for chronic conditions in which participants are randomised to a sequence of interventions. They are a useful and efficient design because participants act as their own control. However, the reporting of crossover trials has been variable and incomplete, which hinders their use in clinical decision making and by future researchers. We present the CONSORT extension to randomised crossover trials, which aims to facilitate better reporting of crossover trials. The CONSORT 2010 checklist is revised for crossover designs, and introduces a modified flowchart and baseline table to enhance transparency. Examples of good reporting and evidence based rationale for CONSORT crossover checklist items are provided.
Figures

References
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
