Cellular Plasmalogen Content Does Not Influence Arachidonic Acid Levels or Distribution in Macrophages: A Role for Cytosolic Phospholipase A2γ in Phospholipid Remodeling
- PMID: 31370188
- PMCID: PMC6721556
- DOI: 10.3390/cells8080799
Cellular Plasmalogen Content Does Not Influence Arachidonic Acid Levels or Distribution in Macrophages: A Role for Cytosolic Phospholipase A2γ in Phospholipid Remodeling
Abstract
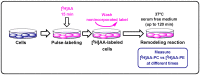
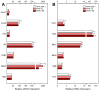
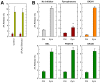
Availability of free arachidonic acid (AA) constitutes a rate limiting factor for cellular eicosanoid synthesis. AA distributes differentially across membrane phospholipids, which is largely due to the action of coenzyme A-independent transacylase (CoA-IT), an enzyme that moves the fatty acid primarily from diacyl phospholipid species to ether-containing species, particularly the ethanolamine plasmalogens. In this work, we examined the dependence of AA remodeling on plasmalogen content using the murine macrophage cell line RAW264.7 and its plasmalogen-deficient variants RAW.12 and RAW.108. All three strains remodeled AA between phospholipids with similar magnitude and kinetics, thus demonstrating that cellular plasmalogen content does not influence the process. Cell stimulation with yeast-derived zymosan also had no effect on AA remodeling, but incubating the cells in AA-rich media markedly slowed down the process. Further, knockdown of cytosolic-group IVC phospholipase A2γ (cPLA2γ) by RNA silencing significantly reduced AA remodeling, while inhibition of other major phospholipase A2 forms such as cytosolic phospholipase A2α, calcium-independent phospholipase A2β, or secreted phospholipase A2 had no effect. These results uncover new regulatory features of CoA-IT-mediated transacylation reactions in cellular AA homeostasis and suggest a hitherto unrecognized role for cPLA2γ in maintaining membrane phospholipid composition via regulation of AA remodeling.
Keywords: arachidonic acid; eicosanoids; inflammation; monocytes/macrophages; phospholipase A2; phospholipid remodeling.
Conflict of interest statement
The authors declare no conflict of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript; or in the decision to publish the results.
Figures






Similar articles
-
Phospholipid Arachidonic Acid Remodeling During Phagocytosis in Mouse Peritoneal Macrophages.Biomedicines. 2020 Aug 5;8(8):274. doi: 10.3390/biomedicines8080274. Biomedicines. 2020. PMID: 32764331 Free PMC article.
-
On the importance of plasmalogen status in stimulated arachidonic acid release in the macrophage cell line RAW 264.7.Biochim Biophys Acta. 2008 Apr;1781(4):213-9. doi: 10.1016/j.bbalip.2008.01.007. Epub 2008 Feb 20. Biochim Biophys Acta. 2008. PMID: 18328831
-
Essential Role for Ethanolamine Plasmalogen Hydrolysis in Bacterial Lipopolysaccharide Priming of Macrophages for Enhanced Arachidonic Acid Release.Front Immunol. 2017 Sep 29;8:1251. doi: 10.3389/fimmu.2017.01251. eCollection 2017. Front Immunol. 2017. PMID: 29033952 Free PMC article.
-
Plasmalogens, phospholipase A2, and docosahexaenoic acid turnover in brain tissue.J Mol Neurosci. 2001 Apr-Jun;16(2-3):263-72; discussion 279-84. doi: 10.1385/jmn:16:2-3:263. J Mol Neurosci. 2001. PMID: 11478381 Review.
-
Lipidomic approaches to the study of phospholipase A2-regulated phospholipid fatty acid incorporation and remodeling.Biochimie. 2010 Jun;92(6):645-50. doi: 10.1016/j.biochi.2009.11.010. Epub 2009 Dec 5. Biochimie. 2010. PMID: 19969040 Review.
Cited by
-
Dynamics of Docosahexaenoic Acid Utilization by Mouse Peritoneal Macrophages.Biomolecules. 2023 Nov 10;13(11):1635. doi: 10.3390/biom13111635. Biomolecules. 2023. PMID: 38002317 Free PMC article.
-
Eicosanoid Metabolomic Profile of Remdesivir Treatment in Rat Plasma by High-Performance Liquid Chromatography Mass Spectrometry.Front Pharmacol. 2021 Sep 29;12:747450. doi: 10.3389/fphar.2021.747450. eCollection 2021. Front Pharmacol. 2021. PMID: 34658883 Free PMC article.
-
Bioactive lipid signaling and lipidomics in macrophage polarization: Impact on inflammation and immune regulation.Front Immunol. 2025 Feb 14;16:1550500. doi: 10.3389/fimmu.2025.1550500. eCollection 2025. Front Immunol. 2025. PMID: 40028333 Free PMC article. Review.
-
Severe SARS-CoV-2 infection in humans is defined by a shift in the serum lipidome resulting in dysregulation of eicosanoid immune mediators.medRxiv [Preprint]. 2020 Jul 13:2020.07.09.20149849. doi: 10.1101/2020.07.09.20149849. medRxiv. 2020. Update in: J Immunol. 2021 Jan 15;206(2):329-334. doi: 10.4049/jimmunol.2001025. PMID: 32676616 Free PMC article. Updated. Preprint.
-
Asymmetric Distribution of Plasmalogens and Their Roles-A Mini Review.Membranes (Basel). 2023 Aug 29;13(9):764. doi: 10.3390/membranes13090764. Membranes (Basel). 2023. PMID: 37755186 Free PMC article. Review.

