Lipid Reshaping and Lipophagy Are Induced in a Modeled Ischemia-Reperfusion Injury of Blood Brain Barrier
- PMID: 31370282
- PMCID: PMC6696511
- DOI: 10.3390/ijms20153752
Lipid Reshaping and Lipophagy Are Induced in a Modeled Ischemia-Reperfusion Injury of Blood Brain Barrier
Abstract
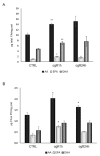
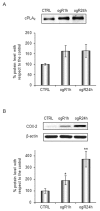
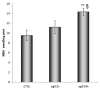
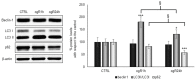
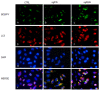
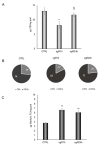
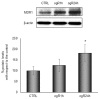
Ischemic-reperfusion (I/R) injury induced a remodeling of protein and lipid homeostasis, under oxidative stress and inflammatory status. Starvation occurring during I/R is a condition leading to autophagy activation, which allows abnormal material clearance or amino acid, or both, and fatty acid (FA) recycling essential for survival. This study investigated the lipid reshaping, peroxidation, and related-signaling pathways, in rat brain endothelial cells (RBE4) subjected to 3 h of oxygen and glucose deprivation (OGD) and restoration of standard condition (I/R in vitro model). Lipids and proteins were analyzed after 1 or 24 h of oxygen and nutrient restoration. Together with the oxidative stress and inflammatory status, I/R injury induced a reshaping of neutral lipids and biogenesis of lipid droplets (LD) with excessive lipid storage. The increase of LC3-II/LC3-I ratio, an autophagy marker, and LC3 co-localization with LD suggest the activation of lipophagy machinery to counteract the cell engulfment. Lipophagy leads to cholesterol ester (CE) hydrolysis, increasing free cholesterol (FC) secretion, which occurred by specific transporters or unconventional exocytosis pathways, or both. Here, we propose that an unconventional spreading of FC and other lipid metabolites may influence the neurovascular unit (NVU) cells, contributing to Blood brain barrier (BBB) alteration or adaptation, or both, to the cumulative effects of several transient ischemia.
Keywords: cholesterol; ischemia; lipid droplets; lipophagy; oxygen and glucose deprivation; vessel disease.
Conflict of interest statement
The authors declare no conflict of interest.
Figures


References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials

