Gadolinium Chloride Restores the Function of the Gap Junctional Intercellular Communication between Hepatocytes in a Liver Injury
- PMID: 31370360
- PMCID: PMC6695937
- DOI: 10.3390/ijms20153748
Gadolinium Chloride Restores the Function of the Gap Junctional Intercellular Communication between Hepatocytes in a Liver Injury
Abstract
Background: Gadolinium chloride (GdCl3) has been reported to attenuate liver injury caused by a variety of toxicants. Gap junctional intercellular communication (GJIC) is thought to be essential in controlling liver homeostasis and pathology. Here we evaluate the effects of GdCl3 on functional GJIC and connexin expression in mouse models and primary hepatocytes.
Methods: Mice were administered GdCl3 intraperitoneally the day before a carbon tetrachloride (CCl4) injection or bile duct ligation (BDL) operation. Primary hepatocytes were treated with CCl4 or lipopolysaccharides (LPS), with or without GdCl3. A scrape loading/dye transfer assay was performed to assess the GJIC function. The expression of connexins was examined by real-time reverse transcription polymerase chain reaction (RT-PCR), western blot and immunofluorescent staining.
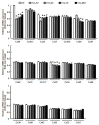
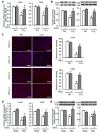
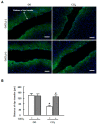
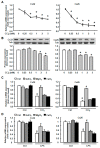
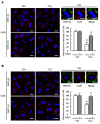
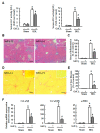
Results: CCl4 treatment or the BDL operation led to the dysfunction of GJIC and a down-regulation of Cx32 and Cx26 in injured liver. GdCl3 administration restored GJIC function between hepatocytes by facilitating the transfer of fluorescent dye from one cell into adjacent cells via GJIC, and markedly prevented the decrease of Cx32 and Cx26 in injured liver. In primary hepatocytes, CCl4 or LPS treatment induced an obvious decline of Cx32 and Cx26, whereas GdCl3 pretreatment prevented the down-regulation of connexins. In vivo GdCl3 protected hepatocytes and attenuated the liver inflammation and fibrosis in liver injury mouse models.
Conclusion: GdCl3 administration protects functional GJIC between hepatocytes, and prevents the decrease of connexin proteins at mRNA and protein levels during liver injury, leading to the alleviation of chronic liver injury.
Keywords: connexin; gadolinium chloride; gap junctional intercellular communication; hepatocyte; liver injury.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

References
-
- Yoshizawa T., Watanabe S., Hirose M., Miyazaki A., Sato N. Dimethylsulfoxide maintains intercellular communication by preserving the gap junctional protein connexin32 in primary cultured hepatocyte doublets from rats. J. Gastroenterol. Hepatol. 1997;12:325–330. doi: 10.1111/j.1440-1746.1997.tb00429.x. - DOI - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

