p120-catenin regulates WNT signaling and EMT in the mouse embryo
- PMID: 31371508
- PMCID: PMC6708312
- DOI: 10.1073/pnas.1902843116
p120-catenin regulates WNT signaling and EMT in the mouse embryo
Abstract
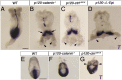
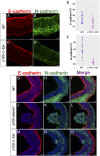
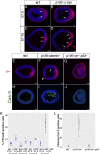
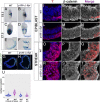
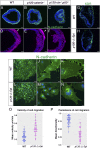
Epithelial-to-mesenchymal transitions (EMTs) require a complete reorganization of cadherin-based cell-cell junctions. p120-catenin binds to the cytoplasmic juxtamembrane domain of classical cadherins and regulates their stability, suggesting that p120-catenin may play an important role in EMTs. Here, we describe the role of p120-catenin in mouse gastrulation, an EMT that can be imaged at cellular resolution and is accessible to genetic manipulation. Mouse embryos that lack all p120-catenin, or that lack p120-catenin in the embryo proper, survive to midgestation. However, mutants have specific defects in gastrulation, including a high rate of p53-dependent cell death, a bifurcation of the posterior axis, and defects in the migration of mesoderm; all are associated with abnormalities in the primitive streak, the site of the EMT. In embryonic day 7.5 (E7.5) mutants, the domain of expression of the streak marker Brachyury (T) expands more than 3-fold, from a narrow strip of posterior cells to encompass more than one-quarter of the embryo. After E7.5, the enlarged T+ domain splits in 2, separated by a mass of mesoderm cells. Brachyury is a direct target of canonical WNT signaling, and the domain of WNT response in p120-catenin mutant embryos, like the T domain, is first expanded, and then split, and high levels of nuclear β-catenin levels are present in the cells of the posterior embryo that are exposed to high levels of WNT ligand. The data suggest that p120-catenin stabilizes the membrane association of β-catenin, thereby preventing accumulation of nuclear β-catenin and excessive activation of the WNT pathway during EMT.
Keywords: WNT signaling; cell migration; epithelial–mesenchymal transition; gastrulation; p53-dependent cell death.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

References
-
- Ishiyama N., et al. , Dynamic and static interactions between p120 catenin and E-cadherin regulate the stability of cell-cell adhesion. Cell 141, 117–128 (2010). - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

