The autism- and schizophrenia-associated protein CYFIP1 regulates bilateral brain connectivity and behaviour
- PMID: 31371726
- PMCID: PMC6672001
- DOI: 10.1038/s41467-019-11203-y
The autism- and schizophrenia-associated protein CYFIP1 regulates bilateral brain connectivity and behaviour
Abstract
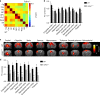
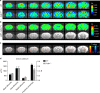
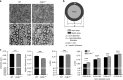
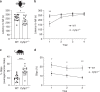
Copy-number variants of the CYFIP1 gene in humans have been linked to autism spectrum disorders (ASD) and schizophrenia (SCZ), two neuropsychiatric disorders characterized by defects in brain connectivity. Here, we show that CYFIP1 plays an important role in brain functional connectivity and callosal functions. We find that Cyfip1-heterozygous mice have reduced functional connectivity and defects in white matter architecture, similar to phenotypes found in patients with ASD, SCZ and other neuropsychiatric disorders. Cyfip1-deficient mice also present decreased myelination in the callosal axons, altered presynaptic function, and impaired bilateral connectivity. Finally, Cyfip1 deficiency leads to abnormalities in motor coordination, sensorimotor gating and sensory perception, which are also known neuropsychiatric disorder-related symptoms. These results show that Cyfip1 haploinsufficiency compromises brain connectivity and function, which might explain its genetic association to neuropsychiatric disorders.
Conflict of interest statement
The authors declare no competing interests.
Figures


Comment in
-
Is There a Hemispheric Disconnect in Neurodevelopmental Disorders?Trends Neurosci. 2019 Dec;42(12):843-844. doi: 10.1016/j.tins.2019.10.005. Epub 2019 Nov 6. Trends Neurosci. 2019. PMID: 31704178
Similar articles
-
Behavioral training rescues motor deficits in Cyfip1 haploinsufficiency mouse model of autism spectrum disorders.Transl Psychiatry. 2019 Jan 21;9(1):29. doi: 10.1038/s41398-018-0338-9. Transl Psychiatry. 2019. PMID: 30664619 Free PMC article.
-
The autism and schizophrenia associated gene CYFIP1 is critical for the maintenance of dendritic complexity and the stabilization of mature spines.Transl Psychiatry. 2014 Mar 25;4(3):e374. doi: 10.1038/tp.2014.16. Transl Psychiatry. 2014. PMID: 24667445 Free PMC article.
-
CYFIP1 Dosages Exhibit Divergent Behavioral Impact via Diametric Regulation of NMDA Receptor Complex Translation in Mouse Models of Psychiatric Disorders.Biol Psychiatry. 2022 Nov 15;92(10):815-826. doi: 10.1016/j.biopsych.2021.04.023. Epub 2021 May 5. Biol Psychiatry. 2022. PMID: 34247782 Free PMC article.
-
Evidence for a Contribution of the Nlgn3/Cyfip1/Fmr1 Pathway in the Pathophysiology of Autism Spectrum Disorders.Neuroscience. 2020 Oct 1;445:31-41. doi: 10.1016/j.neuroscience.2019.10.011. Epub 2019 Nov 6. Neuroscience. 2020. PMID: 31705895 Review.
-
Common mechanisms of excitatory and inhibitory imbalance in schizophrenia and autism spectrum disorders.Curr Mol Med. 2015;15(2):146-67. doi: 10.2174/1566524015666150303003028. Curr Mol Med. 2015. PMID: 25732149 Free PMC article. Review.
Cited by
-
Analysis of Cytogenetic Abnormalities in Iranian Patients with Syndromic Autism Spectrum Disorder: A Case Series.Iran J Child Neurol. 2022 Spring;16(2):117-128. doi: 10.22037/ijcn.v16i4.34843. Epub 2022 Mar 14. Iran J Child Neurol. 2022. PMID: 35497098 Free PMC article.
-
Measuring the Non-linear Directed Information Flow in Schizophrenia by Multivariate Transfer Entropy.Front Comput Neurosci. 2020 Jan 10;13:85. doi: 10.3389/fncom.2019.00085. eCollection 2019. Front Comput Neurosci. 2020. PMID: 31998105 Free PMC article.
-
Dysregulation of the mTOR-FMRP pathway and synaptic plasticity in an environmental model of ASD.Mol Psychiatry. 2025 May;30(5):1937-1951. doi: 10.1038/s41380-024-02805-0. Epub 2024 Nov 27. Mol Psychiatry. 2025. PMID: 39604505 Free PMC article.
-
CYFIP1 overexpression increases fear response in mice but does not affect social or repetitive behavioral phenotypes.Mol Autism. 2019 Jun 7;10:25. doi: 10.1186/s13229-019-0278-0. eCollection 2019. Mol Autism. 2019. PMID: 31198525 Free PMC article.
-
Mapping Cerebellar Morphology in 15q11.2 CNV Carriers Using Normative Modeling.medRxiv [Preprint]. 2025 May 28:2025.05.28.25328353. doi: 10.1101/2025.05.28.25328353. medRxiv. 2025. PMID: 40661298 Free PMC article. Preprint.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases

