Stanniocalcin 2 (STC2) expression promotes post-radiation survival, migration and invasion of nasopharyngeal carcinoma cells
- PMID: 31372045
- PMCID: PMC6636319
- DOI: 10.2147/CMAR.S197607
Stanniocalcin 2 (STC2) expression promotes post-radiation survival, migration and invasion of nasopharyngeal carcinoma cells
Erratum in
-
Erratum: Stanniocalcin 2 (STC2) Expression Promotes Post-Radiation Survival, Migration and Invasion of Nasopharyngeal Carcinoma Cells [Corrigendum].Cancer Manag Res. 2019 Dec 11;11:10409. doi: 10.2147/CMAR.S240959. eCollection 2019. Cancer Manag Res. 2019. PMID: 31853197 Free PMC article.
Abstract
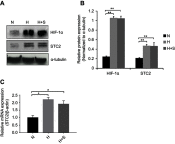
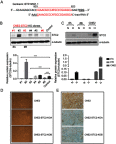
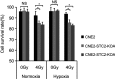
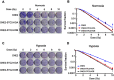
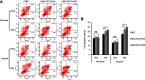
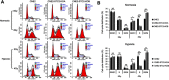
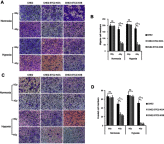
Background: Stanniocalcin 2 (STC2) expression is upregulated under multiple stress conditions including hypoxia, nutrient starvation and radiation. Overexpression of STC2 correlates with tumor progression and poor prognosis. Purpose: We previously demonstrated that overexpression of STC2 in nasopharyngeal carcinomas (NPC) positively correlates with radiation resistance and tumor metastasis, two major clinical obstacles to the improvement of NPC management. However, it remains elusive whether STC2 expression is a critical contributing factor for post-radiation survival and metastasis of NPC cells. Materials and methods: Using the radiation resistant CNE2 cell line as a model, we examined the importance of STC2 expression for post-radiation survival, migration and invasion. Here, we report the establishment of STC2 knockout lines (CNE2-STC2-KO) using the CRISPR/Cas9-based genome editing technique. Results: Compared with the parental line, STC2-KO cells showed similar proliferation and morphology in normal culture conditions, and loss of STC2 did not compromise the cell tumorigenicity in nude mice model. However, STC2-KO lines demonstrated increased sensitivity to X-radiation under either normoxic or hypoxic conditions. Particularly, upon X-radiation, parental CNE2 cells only slightly whereas STC2-KO cells remarkably decreased the migration and invasion ability. Cell cycle analysis revealed that loss of STC2 accumulated cells in G1 and G2/M phases but decreased S-population. Conclusion: These data indicate that the expression of STC2, which can be stimulated by metabolic or therapeutic stresses, is one important factor to promote survival and metastasis of post-radiation NPC cells. Therefore, targeting STC2 or relative downstream pathways may provide novel strategies to overcome radiation resistance and metastasis of NPC.
Keywords: metabolic stress; metastasis; migration; nasopharyngeal carcinoma; radiation resistance; stanniocalcin 2.
Conflict of interest statement
The authors report no conflicts of interest in this work.
Figures
References
-
- Chang AC-M, Janosi J, Hulsbeek M, et al. A novel human cDNA highly homologous to the fish hormone stanniocalcin. Mol Cell Endocrinol. 1995;112(2):241–247. - PubMed
-
- Pacheco J, Vaca L. STIM-TRP Pathways and Microdomain Organization: auxiliary proteins of the STIM/Orai complex Adv Exp Med Biol. 2017:993:189–210. - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

