Predictors of inguinal lymph node metastasis in penile cancer patients: a meta-analysis of retrospective studies
- PMID: 31372046
- PMCID: PMC6628149
- DOI: 10.2147/CMAR.S206579
Predictors of inguinal lymph node metastasis in penile cancer patients: a meta-analysis of retrospective studies
Abstract
Purpose: Inguinal lymph node metastasis (LNM) is one of the most significant prognostic factors for patients with penile cancer. This study aimed to identify potential predictors of inguinal LNM.
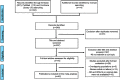
Patients and methods: A comprehensive search of the PubMed, Embase, and Cochrane Library databases for studies that reported predictors of inguinal LNM in penile cancer was performed. Finally, we selected 42 eligible studies with 4,802 patients. Accumulative analyses of odds ratios (ORs) and corresponding 95% confidence intervals (CIs) were performed. All analyses were performed by using Review Manager software version 5.3.
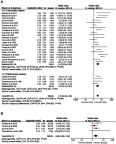
Results: Among the 4,802 patients, 1,706 (36%) were diagnosed with inguinal LNM. Predictors of LNM included two categories: tumor-associated biomarkers and invasive clinicopathologic characteristics. Biomarker-specific predictors: the program death ligand 1 (PD-L1) overexpression (OR=2.55, p=0.002), higher neutrophil-to-lymphocyte ratio (NLR) (OR=4.22, p=0.010), higher C-reactive protein (CRP) (OR=4.78, p<0.001), squamous cell carcinoma antigen (SCC-Ag) overexpression (OR=8.52, p<0.001), P53 protein overexpression (OR=3.57, p<0.001). Clinicopathological predictors: positive clinical lymph node (cN+) (OR=5.86, p<0.001), high-risk histopathological subtype (OR=14.63, p<0.001) and intermediate-risk subtype (OR=3.37, p<0.001), vertical growth pattern (OR=1.97, p=0.020), higher stage (AJCC: OR=3.66, p<0.001; UICC: OR=2.43, p<0.001), higher tumor grade (OR=3.37, p<0.001), tumor size (>3 cm) (OR=2.00, p=0.002), LVI (OR=3.37, p<0.001), invasion depth (>5 mm) (OR=2.58, p=0.002), nerve invasion (OR=2.84, p<0.001), corpora cavernosum invasion (OR=2.22, p<0.001), corpus spongiosum invasion (OR=1.73, p=0.002) and urethra invasion (OR=1.81, p=0.030).
Conclusion: Current meta-analysis conclusively identified valuable predictors of inguinal LNM for patients with penile cancer. However, high-quality studies are warranted to further validate our conclusions. The intrinsic link between these predictors needs to be further investigated to create an accurate mathematical prediction model for LNM.
Keywords: inguinal lymph node metastasis; meta-analysis; penile cancer; predictor.
Conflict of interest statement
The authors report no conflicts of interest in this work.
Figures





References
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

