The Benefit of Reactivating p53 under MAPK Inhibition on the Efficacy of Radiotherapy in Melanoma
- PMID: 31374895
- PMCID: PMC6721382
- DOI: 10.3390/cancers11081093
The Benefit of Reactivating p53 under MAPK Inhibition on the Efficacy of Radiotherapy in Melanoma
Erratum in
-
Correction: Krayem et al. The Benefit of Reactivating p53 under MAPK Inhibition on the Efficacy of Radiotherapy in Melanoma. Cancers 2019, 11, 1093.Cancers (Basel). 2023 Dec 15;15(24):5860. doi: 10.3390/cancers15245860. Cancers (Basel). 2023. PMID: 38136445 Free PMC article.
Abstract
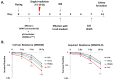
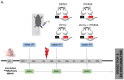
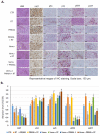
Radiotherapy (RT) in patients with melanoma historically showed suboptimal results, because the disease is often radioresistant due to various mechanisms such as scavenging free radicals by thiols, pigmentary machinery, or enhanced DNA repair. However, radiotherapy has been utilized as adjuvant therapy after the complete excision of primary melanoma and lymph nodes to reduce the rate of nodal recurrences in high-risk patients. The resistance of melanoma cells to radiotherapy may also be in relation with the constitutive activation of the MAPK pathway and/or with the inactivation of p53 observed in about 90% of melanomas. In this study, we aimed to assess the potential benefit of adding RT to BRAF-mutated melanoma cells under a combined p53 reactivation and MAPK inhibition in vitro and in a preclinical animal model. We found that the combination of BRAF inhibition (vemurafenib, which completely shuts down the MAPK pathway), together with p53 reactivation (PRIMA-1Met) significantly enhanced the radiosensitivity of BRAF-mutant melanoma cells. This was accompanied by an increase in both p53 expression and activity. Of note, we found that radiation alone markedly promoted both ERK and AKT phosphorylation, thus contributing to radioresistance. The combination of vemurafenib and PRIMA-1Met caused the inactivation of both MAPK kinase and PI3K/AKT pathways. Furthermore, when combined with radiotherapy, it was able to significantly enhance melanoma cell radiosensitivity. Interestingly, in nude mice bearing melanoma xenografts, the latter triple combination had not only a synergistic effect on tumor growth inhibition, but also a potent control on tumor regrowth in all animals after finishing the triple combination therapy. RT alone had only a weak effect. In conclusion, we provide a basis for a strategy that may overcome the radioresistance of BRAF-mutated melanoma cells to radiotherapy. Whether this will translate into a rational to use radiotherapy in the curative setting in BRAF-mutated melanoma patients deserves consideration.
Keywords: V600EBRAF inhibition; intrinsic and acquired resistance; melanoma; p53 activation; radiotherapy.
Conflict of interest statement
The authors declare no conflict of interest.
Figures



Similar articles
-
p53 Reactivation by PRIMA-1(Met) (APR-246) sensitises (V600E/K)BRAF melanoma to vemurafenib.Eur J Cancer. 2016 Mar;55:98-110. doi: 10.1016/j.ejca.2015.12.002. Epub 2016 Jan 17. Eur J Cancer. 2016. PMID: 26790143
-
A new water soluble MAPK activator exerts antitumor activity in melanoma cells resistant to the BRAF inhibitor vemurafenib.Biochem Pharmacol. 2015 May 1;95(1):16-27. doi: 10.1016/j.bcp.2015.03.004. Epub 2015 Mar 17. Biochem Pharmacol. 2015. PMID: 25795251
-
Overcoming acquired BRAF inhibitor resistance in melanoma via targeted inhibition of Hsp90 with ganetespib.Mol Cancer Ther. 2014 Feb;13(2):353-63. doi: 10.1158/1535-7163.MCT-13-0481. Epub 2014 Jan 7. Mol Cancer Ther. 2014. PMID: 24398428
-
Vemurafenib: targeted inhibition of mutated BRAF for treatment of advanced melanoma and its potential in other malignancies.Drugs. 2012 Dec 3;72(17):2207-22. doi: 10.2165/11640870-000000000-00000. Drugs. 2012. PMID: 23116250 Review.
-
Vemurafenib.Recent Results Cancer Res. 2014;201:215-25. doi: 10.1007/978-3-642-54490-3_13. Recent Results Cancer Res. 2014. PMID: 24756795 Review.
Cited by
-
Targeting RAS-RAF-MEK-ERK signaling pathway in human cancer: Current status in clinical trials.Genes Dis. 2022 May 20;10(1):76-88. doi: 10.1016/j.gendis.2022.05.006. eCollection 2023 Jan. Genes Dis. 2022. PMID: 37013062 Free PMC article. Review.
-
The Emerging Burden of Genetic Instability and Mutation in Melanoma: Role of Molecular Mechanisms.Cancers (Basel). 2022 Dec 15;14(24):6202. doi: 10.3390/cancers14246202. Cancers (Basel). 2022. PMID: 36551688 Free PMC article.
-
Correction: Krayem et al. The Benefit of Reactivating p53 under MAPK Inhibition on the Efficacy of Radiotherapy in Melanoma. Cancers 2019, 11, 1093.Cancers (Basel). 2023 Dec 15;15(24):5860. doi: 10.3390/cancers15245860. Cancers (Basel). 2023. PMID: 38136445 Free PMC article.
-
Kinome Profiling to Predict Sensitivity to MAPK Inhibition in Melanoma and to Provide New Insights into Intrinsic and Acquired Mechanism of Resistance.Cancers (Basel). 2020 Feb 22;12(2):512. doi: 10.3390/cancers12020512. Cancers (Basel). 2020. PMID: 32098410 Free PMC article.
-
Cutaneous Melanoma Classification: The Importance of High-Throughput Genomic Technologies.Front Oncol. 2021 May 28;11:635488. doi: 10.3389/fonc.2021.635488. eCollection 2021. Front Oncol. 2021. PMID: 34123788 Free PMC article. Review.
References
-
- Capasso C., Hirvinen M., Garofalo M., Romaniuk D., Kuryk L., Sarvela T., Vitale A., Antopolsky M., Magarkar A., Viitala T., et al. Oncolytic adenoviruses coated with MHC-I tumor epitopes increase the antitumor immunity and efficacy against melanoma. Oncoimmunology. 2016;5:e1105429. doi: 10.1080/2162402X.2015.1105429. - DOI - PMC - PubMed
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

