1-Trifluoromethoxyphenyl-3-(1-propionylpiperidin-4-yl) Urea, a Selective and Potent Dual Inhibitor of Soluble Epoxide Hydrolase and p38 Kinase Intervenes in Alzheimer's Signaling in Human Nerve Cells
- PMID: 31378059
- PMCID: PMC7028313
- DOI: 10.1021/acschemneuro.9b00271
1-Trifluoromethoxyphenyl-3-(1-propionylpiperidin-4-yl) Urea, a Selective and Potent Dual Inhibitor of Soluble Epoxide Hydrolase and p38 Kinase Intervenes in Alzheimer's Signaling in Human Nerve Cells
Abstract
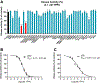
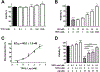
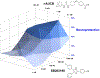
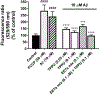
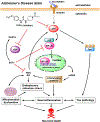
Alzheimer's disease (AD) is the most common neurodegenerative disorder. Neuroinflammation is a prevalent pathogenic stress leading to neuronal death in AD. Targeting neuroinflammation to keep neurons alive is an attractive strategy for AD therapy. 1-Trifluoromethoxyphenyl-3-(1-propionylpiperidin-4-yl) urea (TPPU) is a potent inhibitor of soluble epoxide hydrolase (sEH) and can enter into the brain. It has good efficacy on a wide range of chronic inflammatory diseases in preclinical animal models. However, the anti-neuroinflammatory effects and molecular mechanisms of TPPU for potential AD interventions remain elusive. With an aim to develop multitarget therapeutics for neurodegenerative diseases, we screened TPPU against sEH from different mammalian species and a broad panel of human kinases in vitro for potential new targets relevant to neuroinflammation in AD. TPPU inhibits both human sEH and p38β kinase, two key regulators of inflammation, with nanomolar potencies and distinct selectivity. To further elucidate the molecular mechanisms, differentiated SH-SY5Y human neuroblastoma cells were used as an AD cell model, and we investigated the neuroprotection of TPPU against amyloid oligomers. We found that TPPU effectively prevents neuronal death by mitigating amyloid neurotoxicity, tau hyperphosphorylation, and mitochondrial dysfunction, promoting neurite outgrowth and suppressing activation and nuclear translocation of NF-κB for inflammatory responses in human nerve cells. The results indicate that TPPU is a potent and selective dual inhibitor of sEH and p38β kinase, showing a synergistic action in multiple AD signaling pathways. Our study sheds light upon TPPU and other sEH/p38β dual inhibitors for potential pharmacological interventions in AD.
Keywords: Alzheimer’s disease; dual inhibitor; mitochondrial dysfunction; neuroinflammation; neuroprotection; p38 mitogen-activated protein kinase; soluble epoxide hydrolase.
Conflict of interest statement
Conflict of Interest
B.D.H. is a founder of EicOsis Human Health which is developing sEH inhibitors for treatment of inflammatory and neuropathic pain.
Figures
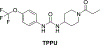


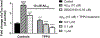
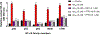
References
-
- Lin MT, and Beal MF (2006) Mitochondrial dysfunction and oxidative stress in neurodegenerative diseases, Nature 443, 787–795. - PubMed
-
- Griffin WST (2013) Neuroinflammatory cytokine signaling and Alzheimer’s disease, N. Engl. J. Med 368, 770–771. - PubMed
-
- Chu D, and Liu F. (2019) Pathological changes of tau related to Alzheimer’s disease, ACS Chem. Neurosci 10, 931–944. - PubMed
-
- Oset-Gasque MJ, and Marco-Contelles J. (2018) Alzheimer’s disease, the “one-molecule, one-target” paradigm, and the multitarget directed ligand approach, ACS Chem. Neurosci 9, 401–403. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

