Structural Engineering of Low-Dimensional Metal-Organic Frameworks: Synthesis, Properties, and Applications
- PMID: 31380160
- PMCID: PMC6662104
- DOI: 10.1002/advs.201802373
Structural Engineering of Low-Dimensional Metal-Organic Frameworks: Synthesis, Properties, and Applications
Abstract
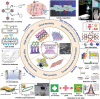
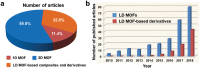
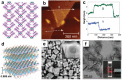
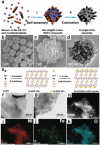
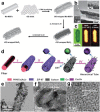
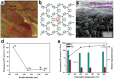
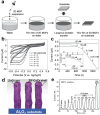
Low-dimensional metal-organic frameworks (LD MOFs) have attracted increasing attention in recent years, which successfully combine the unique properties of MOFs, e.g., large surface area, tailorable structure, and uniform cavity, with the distinctive physical and chemical properties of LD nanomaterials, e.g., high aspect ratio, abundant accessible active sites, and flexibility. Significant progress has been made in the morphological and structural regulation of LD MOFs in recent years. It is still of great significance to further explore the synthetic principles and dimensional-dependent properties of LD MOFs. In this review, recent progress in the synthesis of LD MOF-based materials and their applications are summarized, with an emphasis on the distinctive advantages of LD MOFs over their bulk counterparties. First, the unique physical and chemical properties of LD MOF-based materials are briefly introduced. Synthetic strategies of various LD MOFs, including 1D MOFs, 2D MOFs, and LD MOF-based composites, as well as their derivatives, are then summarized. Furthermore, the potential applications of LD MOF-based materials in catalysis, energy storage, gas adsorption and separation, and sensing are introduced. Finally, challenges and opportunities of this fascinating research field are proposed.
Keywords: catalysts; electrode materials; low‐dimensional nanomaterials; metal–organic frameworks; sensors.
Conflict of interest statement
The authors declare no conflict of interest.
Figures












Similar articles
-
2D Metal-Organic Frameworks: Properties, Synthesis, and Applications in Electrochemical and Optical Biosensors.Biosensors (Basel). 2023 Jan 11;13(1):123. doi: 10.3390/bios13010123. Biosensors (Basel). 2023. PMID: 36671958 Free PMC article. Review.
-
Recent Progress of Low-Dimensional Metal-Organic Frameworks for Aqueous Zinc-Based Batteries.Small. 2024 Sep;20(36):e2402998. doi: 10.1002/smll.202402998. Epub 2024 May 8. Small. 2024. PMID: 38716678 Review.
-
Stable Metal-Organic Frameworks: Design, Synthesis, and Applications.Adv Mater. 2018 Sep;30(37):e1704303. doi: 10.1002/adma.201704303. Epub 2018 Feb 12. Adv Mater. 2018. PMID: 29430732 Review.
-
Two-dimensional metal-organic frameworks and their derivatives for electrochemical energy storage and electrocatalysis.Nanoscale Adv. 2020 Jan 6;2(2):536-562. doi: 10.1039/c9na00719a. eCollection 2020 Feb 18. Nanoscale Adv. 2020. PMID: 36133218 Free PMC article. Review.
-
Site Isolation in Metal-Organic Frameworks Enables Novel Transition Metal Catalysis.Acc Chem Res. 2018 Sep 18;51(9):2129-2138. doi: 10.1021/acs.accounts.8b00297. Epub 2018 Aug 21. Acc Chem Res. 2018. PMID: 30129753
Cited by
-
Advances in Metal-organic Frameworks (MOFs) based Biosensors for Diagnosis: An Update.Curr Top Med Chem. 2022;22(27):2222-2240. doi: 10.2174/1568026622666220829125548. Curr Top Med Chem. 2022. PMID: 36043769
-
Facile one-pot solvothermal preparation of two-dimensional Ni-based metal-organic framework microsheets as a high-performance supercapacitor material.RSC Adv. 2021 Feb 22;11(14):8362-8366. doi: 10.1039/d1ra00259g. eCollection 2021 Feb 17. RSC Adv. 2021. PMID: 35423289 Free PMC article.
-
Application and Development Prospect of Nanoscale Iron Based Metal-Organic Frameworks in Biomedicine.Int J Nanomedicine. 2023 Sep 1;18:4907-4931. doi: 10.2147/IJN.S417543. eCollection 2023. Int J Nanomedicine. 2023. PMID: 37675409 Free PMC article. Review.
-
Highly Efficient Uptake of Cs+ by Robust Layered Metal-Organic Frameworks with a Distinctive Ion Exchange Mechanism.JACS Au. 2022 Jan 20;2(2):492-501. doi: 10.1021/jacsau.1c00533. eCollection 2022 Feb 28. JACS Au. 2022. PMID: 35252998 Free PMC article.
-
Third-Order Optical Nonlinearities of 2D Materials at Telecommunications Wavelengths.Micromachines (Basel). 2023 Jan 25;14(2):307. doi: 10.3390/mi14020307. Micromachines (Basel). 2023. PMID: 36838007 Free PMC article. Review.
References
-
- Furukawa H., Cordova K. E., O'Keeffe M., Yaghi O. M., Science 2013, 341, 1230444. - PubMed
-
- Schneemann A., Bon V., Schwedler I., Senkovska I., Kaskel S., Fischer R. A., Chem. Soc. Rev. 2014, 43, 6062. - PubMed
-
- Zhao W., Peng J., Wang W., Liu S., Zhao Q., Huang W., Coord. Chem. Rev. 2018, 377, 44.
-
- Ding X. J., Jiang H. L., Acc. Chem. Res. 2019, 52, 356. - PubMed
Publication types
LinkOut - more resources
Full Text Sources
Research Materials
