The effect of culture media on large-scale expansion and characteristic of adipose tissue-derived mesenchymal stromal cells
- PMID: 31383013
- PMCID: PMC6683465
- DOI: 10.1186/s13287-019-1331-9
The effect of culture media on large-scale expansion and characteristic of adipose tissue-derived mesenchymal stromal cells
Abstract
Background: Adipose tissue-derived mesenchymal stromal cells (ASCs) have been shown to exhibit some promising properties of their use in regenerative medicine as advanced therapy medicinal products (ATMP). However, different sources of their origin, methods of isolation, and expansion procedures cause the laboratory and clinical results difficult to compare.
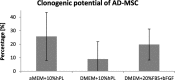
Methods: ASCs were isolated from lipoaspirates and cultured in three different medium formulations: αMEM and DMEM as a basal medium supplemented with 10% of human platelet lysate (hPL) and DMEM supplemented with 20% fetal bovine serum (FBS) and bFGF as a gold standard medium. Subsequently, the impact of culture media on ASCs growth kinetics, their morphology and immunophenotype, ability to differentiate, clonogenic potential, and secretion profile was evaluated.
Results: All cultured ASCs lines showed similar morphology and similar clonogenic potential and have the ability to differentiate into three lines: adipocytes, osteoblasts, and chondroblasts. The immunophenotype of all cultured ASCs was consistent with the guidelines of the International Society for Cell Therapy (ISCT) allowing to define cells as mesenchymal stromal cell (MSC) (≥ 95% CD105, CD73, CD90 and ≤ 2% CD45, CD34, CD14, CD19, HLA-DR). The immunophenotype stabilized after the second passage and did not differ between ASCs cultured in different conditions. The exception was the ASCs grown in the presence of FBS and bFGF, which expressed CD146 antigens. The secretion profile of ASCs cultured in different media was similar. The main secreted cytokine was IL-6, and its level was donor-specific. However, we observed a strong influence of the medium formulation on ASCs growth kinetics. The proliferation rate of ASCs in medium supplemented with hPL was the highest.
Conclusions: Culture media that do not contain animal-derived antigens (xeno-free) can be used to culture cells defined as MSC. Xeno-free medium is a safe alternative for the production of clinical-grade MSC as an advanced therapy medicinal product. Additionally, in such culture conditions, MSC can be easily expanded in accordance with the Good Manufacturing Process (GMP) requirements to a desired amount of cells for clinical applications.
Keywords: Adipose tissue-derived mesenchymal stromal cells; Advanced therapy medicinal products; Human platelet lysate; Interleukin-6.
Conflict of interest statement
The authors declare that they have no competing interests.
Figures





Similar articles
-
Robust differentiation and potent immunomodulation of human mesenchymal stromal cells cultured with a xeno-free GMP protein supplement.Cytotherapy. 2025 Apr;27(4):552-561. doi: 10.1016/j.jcyt.2025.01.003. Epub 2025 Jan 11. Cytotherapy. 2025. PMID: 39864016
-
Comparison of clinical grade human platelet lysates for cultivation of mesenchymal stromal cells from bone marrow and adipose tissue.Scand J Clin Lab Invest. 2016;76(2):93-104. doi: 10.3109/00365513.2015.1099723. Epub 2016 Jan 11. Scand J Clin Lab Invest. 2016. PMID: 26878874
-
Development of fully defined xeno-free culture system for the preparation and propagation of cell therapy-compliant human adipose stem cells.Stem Cell Res Ther. 2013 Mar 7;4(2):27. doi: 10.1186/scrt175. Stem Cell Res Ther. 2013. PMID: 23497764 Free PMC article.
-
Systematic review and meta-analysis on the use of human platelet lysate for mesenchymal stem cell cultures: comparison with fetal bovine serum and considerations on the production protocol.Stem Cell Res Ther. 2022 Apr 4;13(1):142. doi: 10.1186/s13287-022-02815-1. Stem Cell Res Ther. 2022. PMID: 35379348 Free PMC article.
-
Making the Switch: Alternatives to Fetal Bovine Serum for Adipose-Derived Stromal Cell Expansion.Front Cell Dev Biol. 2016 Oct 17;4:115. doi: 10.3389/fcell.2016.00115. eCollection 2016. Front Cell Dev Biol. 2016. PMID: 27800478 Free PMC article. Review.
Cited by
-
Human Mesenchymal Stem Cell (hMSC) Donor Potency Selection for the "First in Cystic Fibrosis" Phase I Clinical Trial (CEASE-CF).Pharmaceuticals (Basel). 2023 Feb 1;16(2):220. doi: 10.3390/ph16020220. Pharmaceuticals (Basel). 2023. PMID: 37259368 Free PMC article.
-
Cell Sources for Human In vitro Bone Models.Curr Osteoporos Rep. 2021 Feb;19(1):88-100. doi: 10.1007/s11914-020-00648-6. Epub 2021 Jan 15. Curr Osteoporos Rep. 2021. PMID: 33447910 Free PMC article. Review.
-
Human mesenchymal stem cell secretomes: Factors affecting profiling and challenges in clinical application.Cell Tissue Res. 2024 Mar;395(3):227-250. doi: 10.1007/s00441-023-03857-4. Epub 2024 Jan 20. Cell Tissue Res. 2024. PMID: 38244032 Review.
-
Effects of adipose-derived mesenchymal stem cell conditioned medium on human tenocytes exposed to high glucose.Ther Adv Musculoskelet Dis. 2024 Jan 8;16:1759720X231214903. doi: 10.1177/1759720X231214903. eCollection 2024. Ther Adv Musculoskelet Dis. 2024. PMID: 38204801 Free PMC article.
-
Large-Scale Expansion of Porcine Adipose-Derived Stem Cells Based on Microcarriers System for Cultured Meat Production.Foods. 2022 Oct 26;11(21):3364. doi: 10.3390/foods11213364. Foods. 2022. PMID: 36359977 Free PMC article.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

