Intragenic antimicrobial peptides (IAPs) from human proteins with potent antimicrobial and anti-inflammatory activity
- PMID: 31386688
- PMCID: PMC6684085
- DOI: 10.1371/journal.pone.0220656
Intragenic antimicrobial peptides (IAPs) from human proteins with potent antimicrobial and anti-inflammatory activity
Abstract
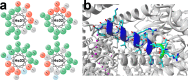
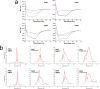
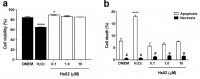
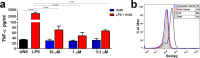
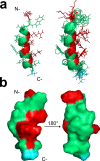
Following the treads of our previous works on the unveiling of bioactive peptides encrypted in plant proteins from diverse species, the present manuscript reports the occurrence of four proof-of-concept intragenic antimicrobial peptides in human proteins, named Hs IAPs. These IAPs were prospected using the software Kamal, synthesized by solid phase chemistry, and had their interactions with model phospholipid vesicles investigated by differential scanning calorimetry and circular dichroism. Their antimicrobial activity against bacteria, yeasts and filamentous fungi was determined, along with their cytotoxicity towards erythrocytes. Our data demonstrates that Hs IAPs are capable to bind model membranes while attaining α-helical structure, and to inhibit the growth of microorganisms at concentrations as low as 1μM. Hs02, a novel sixteen residue long internal peptide (KWAVRIIRKFIKGFIS-NH2) derived from the unconventional myosin 1h protein, was further investigated in its capacity to inhibit lipopolysaccharide-induced release of TNF-α in murine macrophages. Hs02 presented potent anti-inflammatory activity, inhibiting the release of TNF-α in LPS-primed cells at the lowest assayed concentration, 0.1 μM. A three-dimensional solution structure of Hs02 bound to DPC micelles was determined by Nuclear Magnetic Resonance. Our work exemplifies how the human genome can be mined for molecules with biotechnological potential in human health and demonstrates that IAPs are actual alternatives to antimicrobial peptides as pharmaceutical agents or in their many other putative applications.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures
Similar articles
-
Characterization of novel human intragenic antimicrobial peptides, incorporation and release studies from ureasil-polyether hybrid matrix.Mater Sci Eng C Mater Biol Appl. 2021 Feb;119:111581. doi: 10.1016/j.msec.2020.111581. Epub 2020 Oct 1. Mater Sci Eng C Mater Biol Appl. 2021. PMID: 33321627
-
Antimicrobial and anti-inflammatory activities of three chensinin-1 peptides containing mutation of glycine and histidine residues.Sci Rep. 2017 Jan 5;7:40228. doi: 10.1038/srep40228. Sci Rep. 2017. PMID: 28054660 Free PMC article.
-
Rational design of tryptophan-rich antimicrobial peptides with enhanced antimicrobial activities and specificities.Chembiochem. 2010 Nov 2;11(16):2273-82. doi: 10.1002/cbic.201000372. Chembiochem. 2010. PMID: 20865718
-
Solution NMR studies of amphibian antimicrobial peptides: linking structure to function?Biochim Biophys Acta. 2009 Aug;1788(8):1639-55. doi: 10.1016/j.bbamem.2009.01.002. Epub 2009 Jan 15. Biochim Biophys Acta. 2009. PMID: 19272309 Review.
-
Manufacturing of peptides exhibiting biological activity.Amino Acids. 2013 Feb;44(2):315-20. doi: 10.1007/s00726-012-1379-7. Epub 2012 Aug 23. Amino Acids. 2013. PMID: 22914979 Free PMC article. Review.
Cited by
-
Release of immunomodulatory peptides at bacterial membrane interfaces as a novel strategy to fight microorganisms.J Biol Chem. 2023 Apr;299(4):103056. doi: 10.1016/j.jbc.2023.103056. Epub 2023 Feb 22. J Biol Chem. 2023. PMID: 36822328 Free PMC article.
-
Antimicrobial peptide Hs02 with rapid bactericidal, anti-biofilm, and anti-inflammatory activity against carbapenem-resistant Klebsiella pneumoniae and Escherichia coli.Microbiol Spectr. 2025 Jan 7;13(1):e0105024. doi: 10.1128/spectrum.01050-24. Epub 2024 Dec 3. Microbiol Spectr. 2025. PMID: 39625293 Free PMC article.
-
In Silico and In Vitro Analyses Reveal Promising Antimicrobial Peptides from Myxobacteria.Probiotics Antimicrob Proteins. 2023 Feb;15(1):202-214. doi: 10.1007/s12602-022-10036-4. Epub 2022 Dec 31. Probiotics Antimicrob Proteins. 2023. PMID: 36586039 Free PMC article.
-
Intragenic Antimicrobial Peptide Hs02 Hampers the Proliferation of Single- and Dual-Species Biofilms of P. aeruginosa and S. aureus: A Promising Agent for Mitigation of Biofilm-Associated Infections.Int J Mol Sci. 2019 Jul 23;20(14):3604. doi: 10.3390/ijms20143604. Int J Mol Sci. 2019. PMID: 31340580 Free PMC article.
-
Neuroprotective effects on microglia and insights into the structure-activity relationship of an antioxidant peptide isolated from Pelophylax perezi.J Cell Mol Med. 2022 May;26(10):2793-2807. doi: 10.1111/jcmm.17292. Epub 2022 Apr 23. J Cell Mol Med. 2022. PMID: 35460166 Free PMC article.
References
-
- Brand GD. Estratégias para prospecção e predição de peptídeos bioativos [Internet]. Universidade de Brasília. 2007. Available: http://repositorio.unb.br/handle/10482/1373
-
- Phelan M, Aherne A, FitzGerald RJ, O’Brien NM. Casein-derived bioactive peptides: Biological effects, industrial uses, safety aspects and regulatory status. Int Dairy J. Elsevier; 2009;19: 643–654. 10.1016/J.IDAIRYJ.2009.06.001 - DOI
-
- Dale CS, Pagano RDL, Rioli V. Hemopressin: a novel bioactive peptide derived from the alpha1-chain of hemoglobin. Mem Inst Oswaldo Cruz. 2005;100 Suppl: 105–6. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

