HER2 double-equivocal breast cancer in Chinese patients: a high concordance of HER2 status between different blocks from the same tumor
- PMID: 31388934
- PMCID: PMC6797640
- DOI: 10.1007/s10549-019-05387-6
HER2 double-equivocal breast cancer in Chinese patients: a high concordance of HER2 status between different blocks from the same tumor
Abstract
Purpose: Human epidermal growth factor receptor 2 (HER2) status is both an independent prognostic factor and a predictive factor for the efficacy of targeted therapy for breast cancer, so it is critical to accurately detect HER2 protein expression and/or gene amplification. According to the recommendations of the 2013 American Society of Clinical Oncology and College of American Pathologists (ASCO/CAP) guidelines for HER2 breast cancer testing, an additional test should be pursued on a different block from the same tumor as one of the options for patients with immunohistochemistry (IHC) 2+ and a HER2/CEP17 ratio of < 2.0 with an average HER2 signals per tumor cell of ≥ 4.0 and < 6.0 by reflex test using dual-probe fluorescence in situ hybridization (FISH) (double-equivocal HER2). Our aim in this study is to explore the consistency of HER2 status between the two blocks.
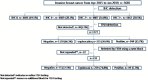
Methods: We retrospectively analyzed 5685 primary invasive breast cancers between April 2015 and January 2019 from Peking Union Medical College Hospital. For cases with double-equivocal HER2 revealed in initial blocks, HER2 gene status was evaluated by FISH in a different block from the same tumor. The FISH score was interpreted according to the 2013 ASCO/CAP guidelines for HER2 testing.
Results: In our cohort of 5685 specimens, the overall HER2 IHC3+, 2+, 1+ and 0 cases were 20.5%, 31.8%, 28.3%, and 19.5%, respectively. Then, 13.7%, 66.3%, and 20.0% of HER2 amplification, non-amplification, and equivocation rates were found, respectively, in IHC2+ patients (n = 1777) by reflex FISH assay. For specimens with double-equivocal HER2 (n = 333), HER2 status was assessed in another block from the same tumor by FISH and then the frequency of HER2 positive, negative, and equivocation was estimated at 5.7%, 22.5%, and 71.8%, respectively. Because double-equivocal HER2 cases are classified in the HER2 negative category by the 2018 ASCO/CAP HER2 testing guidelines, only 1.3% (19/1511) of HER2 positive patients were determined through additional HER2 testing in another block from the HER2 negative population.
Conclusions: HER2 status in different blocks from the same tumor in primary invasive breast cancer was highly concordant. Our data supported the recommendation of the 2018 ASCO/CAP HER2 testing guidelines in breast cancer to remove the suggestion for additional HER2 testing using another block contained in the previous version.
Keywords: Breast cancer; Equivocal; FISH; HER2; IHC.
Conflict of interest statement
All authors declare that they have no conflicts of interest.
Figures


References
-
- Ross JS, Gray GS. Targeted therapy for cancer: the HER-2/neu and herceptin story. Clin Leadersh Manag Rev. 2003;17(6):333–340. - PubMed
-
- Wolff AC, Hammond ME, Hicks DG, Dowsett M, McShane LM, Allison KH, Allred DC, Bartlett JM, Bilous M, Fitzgibbons P, Hanna W, Jenkins RB, Mangu PB, Paik S, Perez EA, Press MF, Spears PA, Vance GH, Viale G, Hayes DF, American Society of Clinical Oncology; College of American Pathologists Recommendations for human epidermal growth factor receptor 2 testing in breast cancer: American Society of Clinical Oncology/College of American Pathologists clinical practice guideline update. J Clin Oncol. 2013;31(31):3997–4013. doi: 10.1200/jco.2013.50.9984. - DOI - PubMed
MeSH terms
Substances
Grants and funding
- 2017YFC1309004/National Key Research and Development Program of China
- 2016-I2M-1-002/Chinese Academy of Medical Sciences (CAMS) Initiative for Innovative Medicine
- 2017-I2M-1-005/Chinese Academy of Medical Sciences (CAMS) Initiative for Innovative Medicine
- 2015KJRK1L01/National Human and Population Scientific Data Sharing Platform
- 2016ZX310176-2/Pathology Research Center of the Chinese Academy of Medical Sciences
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

