Nocebo in Biosimilars and Generics in Neurology: A Systematic Review
- PMID: 31396084
- PMCID: PMC6667943
- DOI: 10.3389/fphar.2019.00809
Nocebo in Biosimilars and Generics in Neurology: A Systematic Review
Abstract
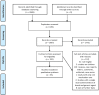
Background: Nocebo refers to adverse events related to patients' negative expectations and previous experiences, mediated by several neurobiological pathways within the brain. It is common among neurological patients and affects adherence and treatment outcomes, representing a real clinical challenge. Methods: We conducted a systematic search based on the PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) guidelines in MEDLINE database, using several keywords for studies that can be processed to investigate the magnitude of nocebo in generics and biosimilars used in the most common neurological diseases. The aim was to estimate its size and suggest strategies to minimize its prevalence in clinical trials and practice. Results: Of a total of 2,606 identified articles, after criteria-based selection, 35 studies were included for analysis. Overall, there was vast heterogeneity across the studies concerning population, study design, and outcomes. Nocebo response could be estimated only in one double-blind randomized trial of generic glatiramer acetate in relapsing remitting multiple sclerosis that included a placebo arm. In this trial, no significant differences observed between the three arms (innovator, bioequivalent, and placebo) in favorable and unfavorable outcomes. In the open-label phase of the trial, an increased withdrawal rate was recorded in patients switched from placebo to bioequivalent (8.4%) that may be related to nocebo. In other open-label and real-world studies evaluating biosimilars or generics for brain disorders, a similar indirect nocebo effect is assuming by several investigators. Also, knowledge gaps between health-care providers and patients exist towards generics and biosimilars. Conclusions: Despite its presence, the true burden of the nocebo response and effect cannot be accurately estimated in existing studies with generics and biosimilars in neurological diseases. Targeted strategies for clinical trials' design are needed in order to measure the exact nocebo's size.
Keywords: biosimilars; generics; neurological diseases; nocebo; nocebo effect; nocebo response.
Figures
References
-
- Abolfazli R., Hosseini A., Gholami K., Javadi M. R., Torkamandi H., Emami S. (2012). Quality of life assessment in patients with multiple sclerosis receiving interferon beta-1a: a comparative longitudinal study of avonex and its biosimilar CinnoVex. ISRN Neurol. 2012, 786526. 10.5402/2012/786526 - DOI - PMC - PubMed
-
- Agency EM Guideline on similar biological medicinal products. EMA; London, UK: (2005).
Publication types
LinkOut - more resources
Full Text Sources