Abnormal fatty acid metabolism is a core component of spinal muscular atrophy
- PMID: 31402618
- PMCID: PMC6689695
- DOI: 10.1002/acn3.50855
Abnormal fatty acid metabolism is a core component of spinal muscular atrophy
Abstract
Objective: Spinal muscular atrophy (SMA) is an inherited neuromuscular disorder leading to paralysis and subsequent death in young children. Initially considered a motor neuron disease, extra-neuronal involvement is increasingly recognized. The primary goal of this study was to investigate alterations in lipid metabolism in SMA patients and mouse models of the disease.
Methods: We analyzed clinical data collected from a large cohort of pediatric SMA type I-III patients as well as SMA type I liver necropsy data. In parallel, we performed histology, lipid analysis, and transcript profiling in mouse models of SMA.
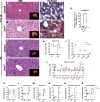
Results: We identify an increased susceptibility to developing dyslipidemia in a cohort of 72 SMA patients and liver steatosis in pathological samples. Similarly, fatty acid metabolic abnormalities were present in all SMA mouse models studied. Specifically, Smn2B/- mice displayed elevated hepatic triglycerides and dyslipidemia, resembling non-alcoholic fatty liver disease (NAFLD). Interestingly, this phenotype appeared prior to denervation.
Interpretation: This work highlights metabolic abnormalities as an important feature of SMA, suggesting implementation of nutritional and screening guidelines in patients, as such defects are likely to increase metabolic distress and cardiovascular risk. This study emphasizes the need for a systemic therapeutic approach to ensure maximal benefits for all SMA patients throughout their life.
© 2019 The Authors. Annals of Clinical and Translational Neurology published by Wiley Periodicals, Inc on behalf of American Neurological Association.
Conflict of interest statement
Marc‐Olivier Deguise received honoraria and travel accommodations by Biogen for the SMA Summit 2018 held in Montreal, Canada. Rashmi Kothary and the Ottawa Hospital Research Institute have a licensing agreement with Biogen for the
Figures




References
-
- Lefebvre S, Burglen L, Reboullet S, et al. Identification and characterization of a spinal muscular atrophy‐ determining gene. Cell 1995;80:155–165. - PubMed
-
- Tisdale S, Pellizzoni L.RNA‐processing dysfunction in spinal muscular atrophy In: Sumner CJ, Paushkin S, C. Ko CP, eds. Spinal muscular atrophy: disease mechanisms and therapy. pp. 113–131. London: Academic Press, 2017.
-
- Shafey D, Cote PD, Kothary R. Hypomorphic Smn knockdown C2C12 myoblasts reveal intrinsic defects in myoblast fusion and myotube morphology. Exp Cell Res 2005;311:49–61. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
