Vanishing white matter: deregulated integrated stress response as therapy target
- PMID: 31402619
- PMCID: PMC6689685
- DOI: 10.1002/acn3.50826
Vanishing white matter: deregulated integrated stress response as therapy target
Abstract
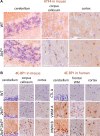
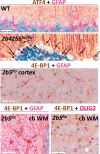
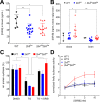
Objective: Vanishing white matter (VWM) is a fatal, stress-sensitive leukodystrophy that mainly affects children and is currently without treatment. VWM is caused by recessive mutations in eukaryotic initiation factor 2B (eIF2B) that is crucial for initiation of mRNA translation and its regulation during the integrated stress response (ISR). Mutations reduce eIF2B activity. VWM pathomechanisms remain unclear. In contrast with the housekeeping function of eIF2B, astrocytes are selectively affected in VWM. One study objective was to test our hypothesis that in the brain translation of specific mRNAs is altered by eIF2B mutations, impacting primarily astrocytes. The second objective was to investigate whether modulation of eIF2B activity could ameliorate this altered translation and improve the disease.
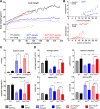
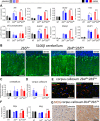
Methods: Mice with biallelic missense mutations in eIF2B that recapitulate human VWM were used to screen for mRNAs with altered translation in brain using polysomal profiling. Findings were verified in brain tissue from VWM patients using qPCR and immunohistochemistry. The compound ISRIB (for "ISR inhibitor") was administered to VWM mice to increase eIF2B activity. Its effect on translation, neuropathology, and clinical signs was assessed.
Results: In brains of VWM compared to wild-type mice we observed the most prominent changes in translation concerning ISR mRNAs; their expression levels correlated with disease severity. We substantiated these findings in VWM patients' brains. ISRIB normalized expression of mRNA markers, ameliorated brain white matter pathology and improved motor skills in VWM mice.
Interpretation: The present findings show that ISR deregulation is central in VWM pathomechanisms and a viable target for therapy.
© 2019 The Authors. Annals of Clinical and Translational Neurology published by Wiley Periodicals, Inc on behalf of American Neurological Association.
Conflict of interest statement
MSvdK and TEMA have a patent PCT/NL2018/050293 on Guanabenz in VWM pending. Otherwise, the authors have declared that no competing interest exists.
Figures


References
-
- Liu R, van der Lei HD, Wang X, et al. Severity of vanishing white matter disease does not correlate with deficits in eIF2B activity or the integrity of eIF2B complexes. Hum Mutat 2011;32:1036–1045. - PubMed
-
- Leegwater PA, Vermeulen G, Konst AA, et al. Subunits of the translation initiation factor eIF2B are mutant in leukoencephalopathy with vanishing white matter. Nat Genet 2001;29:383–388. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

