ROR1-CAR T cells are effective against lung and breast cancer in advanced microphysiologic 3D tumor models
- PMID: 31415244
- PMCID: PMC6795380
- DOI: 10.1172/jci.insight.126345
ROR1-CAR T cells are effective against lung and breast cancer in advanced microphysiologic 3D tumor models
Abstract
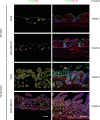
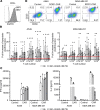
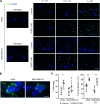
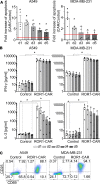
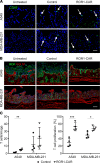
Solid tumors impose immunologic and physical barriers to the efficacy of chimeric antigen receptor (CAR) T cell therapy that are not reflected in conventional preclinical testing against singularized tumor cells in 2-dimensional culture. Here, we established microphysiologic three-dimensional (3D) lung and breast cancer models that resemble architectural and phenotypical features of primary tumors and evaluated the antitumor function of receptor tyrosine kinase-like orphan receptor 1-specific (ROR1-specific) CAR T cells. 3D tumors were established from A549 (non-small cell lung cancer) and MDA-MB-231 (triple-negative breast cancer) cell lines on a biological scaffold with intact basement membrane (BM) under static and dynamic culture conditions, which resulted in progressively increasing cell mass and invasive growth phenotype (dynamic > static; MDA-MB-231 > A549). Treatment with ROR1-CAR T cells conferred potent antitumor effects. In dynamic culture, CAR T cells actively entered arterial medium flow and adhered to and infiltrated the tumor mass. ROR1-CAR T cells penetrated deep into tumor tissue and eliminated multiple layers of tumor cells located above and below the BM. The microphysiologic 3D tumor models developed in this study are standardized, scalable test systems that can be used either in conjunction with or in lieu of animal testing to interrogate the antitumor function of CAR T cells and to obtain proof of concept for their safety and efficacy before clinical application.
Keywords: Breast cancer; Immunology; Immunotherapy; Lung cancer; Oncology.
Conflict of interest statement
Figures

Similar articles
-
Inhibition of TGF-β-receptor signaling augments the antitumor function of ROR1-specific CAR T-cells against triple-negative breast cancer.J Immunother Cancer. 2020 Apr;8(1):e000676. doi: 10.1136/jitc-2020-000676. J Immunother Cancer. 2020. PMID: 32303620 Free PMC article.
-
Immunogenic Chemotherapy Enhances Recruitment of CAR-T Cells to Lung Tumors and Improves Antitumor Efficacy when Combined with Checkpoint Blockade.Cancer Cell. 2021 Feb 8;39(2):193-208.e10. doi: 10.1016/j.ccell.2020.11.005. Epub 2020 Dec 24. Cancer Cell. 2021. PMID: 33357452 Free PMC article.
-
Receptor affinity and extracellular domain modifications affect tumor recognition by ROR1-specific chimeric antigen receptor T cells.Clin Cancer Res. 2013 Jun 15;19(12):3153-64. doi: 10.1158/1078-0432.CCR-13-0330. Epub 2013 Apr 25. Clin Cancer Res. 2013. PMID: 23620405 Free PMC article.
-
Chimeric antigen receptor-engineered T-cell therapy for liver cancer.Hepatobiliary Pancreat Dis Int. 2018 Aug;17(4):301-309. doi: 10.1016/j.hbpd.2018.05.005. Epub 2018 May 24. Hepatobiliary Pancreat Dis Int. 2018. PMID: 29861325 Review.
-
CAR T-cell therapy for triple-negative breast cancer: Where we are.Cancer Lett. 2020 Oct 28;491:121-131. doi: 10.1016/j.canlet.2020.07.044. Epub 2020 Aug 12. Cancer Lett. 2020. PMID: 32795486 Review.
Cited by
-
Efficacy, Safety, and Challenges of CAR T-Cells in the Treatment of Solid Tumors.Cancers (Basel). 2022 Dec 3;14(23):5983. doi: 10.3390/cancers14235983. Cancers (Basel). 2022. PMID: 36497465 Free PMC article. Review.
-
CAR-T cell therapy for lung cancer: Potential and perspective.Thorac Cancer. 2022 Apr;13(7):889-899. doi: 10.1111/1759-7714.14375. Epub 2022 Mar 15. Thorac Cancer. 2022. PMID: 35289077 Free PMC article. Review.
-
Anti-ROR1 CAR-T cells: Architecture and performance.Front Med (Lausanne). 2023 Feb 17;10:1121020. doi: 10.3389/fmed.2023.1121020. eCollection 2023. Front Med (Lausanne). 2023. PMID: 36873868 Free PMC article. Review.
-
Developing ROR1 Targeting CAR-T Cells against Solid Tumors in Preclinical Studies.Cancers (Basel). 2022 Jul 25;14(15):3618. doi: 10.3390/cancers14153618. Cancers (Basel). 2022. PMID: 35892876 Free PMC article.
-
Recent Progress on Immunotherapy for Breast Cancer: Tumor Microenvironment, Nanotechnology and More.Front Bioeng Biotechnol. 2021 Jun 2;9:680315. doi: 10.3389/fbioe.2021.680315. eCollection 2021. Front Bioeng Biotechnol. 2021. PMID: 34150736 Free PMC article. Review.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

