Novel method for action potential measurements from intact cardiac monolayers with multiwell microelectrode array technology
- PMID: 31417144
- PMCID: PMC6695445
- DOI: 10.1038/s41598-019-48174-5
Novel method for action potential measurements from intact cardiac monolayers with multiwell microelectrode array technology
Abstract
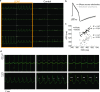
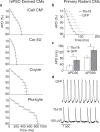
The cardiac action potential (AP) is vital for understanding healthy and diseased cardiac biology and drug safety testing. However, techniques for high throughput cardiac AP measurements have been limited. Here, we introduce a novel technique for reliably increasing the coupling of cardiomyocyte syncytium to planar multiwell microelectrode arrays, resulting in a stable, label-free local extracellular action potential (LEAP). We characterized the reliability and stability of LEAP, its relationship to the field potential, and its efficacy for quantifying AP morphology of human induced pluripotent stem cell derived and primary rodent cardiomyocytes. Rise time, action potential duration, beat period, and triangulation were used to quantify compound responses and AP morphology changes induced by genetic modification. LEAP is the first high throughput, non-invasive, label-free, stable method to capture AP morphology from an intact cardiomyocyte syncytium. LEAP can accelerate our understanding of stem cell models, while improving the automation and accuracy of drug testing.
Conflict of interest statement
H.B.H., A.M.N., C.A.A., S.A.C., D.D.S., M.C., J.D.R. and D.C.M. are employees of Axion BioSystems Inc.
Figures






References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

