Status epilepticus in patients with genetic (idiopathic) generalized epilepsy
- PMID: 31417260
- PMCID: PMC6593751
- DOI: 10.2147/NDT.S209084
Status epilepticus in patients with genetic (idiopathic) generalized epilepsy
Abstract
Aim of the study: Genetic (idiopathic) generalized epilepsies (GGEs) account for nearly one-third of all epilepsies. The frequency of status epilepticus (SE) in patients with GGEs has been poorly studied. Therefore, this study aimed to evaluate the frequency of different forms of SE in a cohort of patients with GGEs.
Materials and methods: Among 153 patients with GGEs treated at the university epilepsy clinic in the period between 1998 and 2018, those with SE were retrospectively identified.
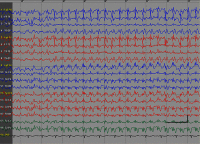
Results: Absence SE was diagnosed in 8 patients (13 episodes), while myoclonic SE was found in 2 patients (2 episodes). No cases of tonic-clonic SE were detected in the study cohort. Most SE episodes were found to be provoked by ill-advised antiepileptic drugs or changes in drug regimen. In all the subjects, SE was stopped by intravenous administration of diazepam and/or valproate. Long-term outcome of epilepsy was good, with most patients (70%) being seizure-free.
Conclusion: Status epilepticus is not a rare phenomenon in patients with genetic generalized epilepsies, with absence SE being the most common type. Most cases of SE are provoked by ill-advised AEDs or changes in drug regimen. Status epilepticus in GGEs can be easily treated with benozdiazepines and/or valproate. Status epilepticus in GGEs can be easily treated with benozdiazepines and/or valproate.
Keywords: absence status epilepticus; genetic generalized epilepsy; myoclonic status epilepticus.
Conflict of interest statement
MB received honoraria for publications and the participation in advisory meetings from Sanofi; honoraria for lectures, travel expenses and conference fees from Sanofi, Adamed, Teva Pharmaceutical, Neuraxpharm, Glenmark and UCB Pharma. AS received honoraria for lectures from Bayer, Boehringer Ingelheim, Novartis, Polpharma, Bristol-Myers Squibb, Novartis, Biogen, Teva Pharmaceutical, Medtronic and for the participation in advisory meetings from Bayer, Boehringer Ingelheim and Novartis. The authors report no other conflicts of interest in this work.
Figures
References
LinkOut - more resources
Full Text Sources
Research Materials