Does Pioglitazone Lead to Neutrophil Extracellular Traps Formation in Chronic Granulomatous Disease Patients?
- PMID: 31428088
- PMCID: PMC6689990
- DOI: 10.3389/fimmu.2019.01739
Does Pioglitazone Lead to Neutrophil Extracellular Traps Formation in Chronic Granulomatous Disease Patients?
Abstract
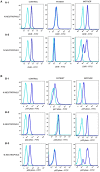
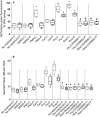
Nicotinamide adenine dinucleotide phosphate (NADPH) oxidase, the enzyme complex responsible for reactive oxygen species (ROS) production, is defective in chronic granulomatous disease (CGD) patients. This enzyme helps in antimicrobial host defense by phagocytes. CGD patients are unable to form neutrophil extracellular traps (NETs), which are composed of granule-derived proteins from neutrophils decorated with decondensed chromatin. Mitochondria have gained attention, being a rich source of flavochrome enzymes due to the presence of several sites for superoxide production. Recently, PPARγ agonists, a mitochondrial ROS inducer, induce mitochondrial ROS formation post-treatment in murine NADPH oxidase knockout models. Mitochondrial ROS is also essential for NOX-independent NETosis. Our study for the first time detects induction of NETosis independent of NADPH oxidase post-treatment with agonists such as pioglitazone and rosiglitazone in CGD subjects. Neutrophils isolated from CGD subjects were treated with pioglitazone and rosiglitazone. After treatment, qualitative analysis of NET formation was done using confocal microscopy after staining with DAPI. Quantitative estimation of extracellular DNA was performed using Sytox green. Mitochondrial ROS production with PPARγ agonist-treated/untreated neutrophils was detected using MitoSOX red. Pioglitazone and rosiglitazone induce significant NET formation in CGD patients. Our data clearly signify the effect of PPARγ agonists in induction of NET formation in CGD cases. Apart from the proposed experimental studies regarding the detailed mechanism of action, controlled trials could provide valuable information regarding the clinical use of pioglitazone in CGD patients as curative HSCT remains challenging in developing countries.
Keywords: MitoSOX red; NOX independnent NETosis; chronic granulomatous disease; mitochondrial ROS; neutrophil extracellular traps; peroxisome proliferator-activated receptor gamma agonists; pioglitazone; reactive oxygen species.
Figures

References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

