Myo-inositol oxygenase expression profile modulates pathogenic ferroptosis in the renal proximal tubule
- PMID: 31437128
- PMCID: PMC6819105
- DOI: 10.1172/JCI129903
Myo-inositol oxygenase expression profile modulates pathogenic ferroptosis in the renal proximal tubule
Abstract
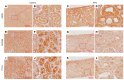
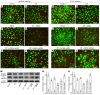
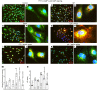
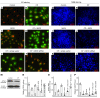
Overexpression of myo-inositol oxygenase (MIOX), a proximal tubular enzyme, exacerbates cellular redox injury in acute kidney injury (AKI). Ferroptosis, a newly coined term associated with lipid hydroperoxidation, plays a critical role in the pathogenesis of AKI. Whether or not MIOX exacerbates tubular damage by accelerating ferroptosis in cisplatin-induced AKI remains elusive. Cisplatin-treated HK-2 cells exhibited notable cell death, which was reduced by ferroptosis inhibitors. Also, alterations in various ferroptosis metabolic sensors, including lipid hydroperoxidation, glutathione peroxidase 4 (GPX4) activity, NADPH and reduced glutathione (GSH) levels, and ferritinophagy, were observed. These perturbations were accentuated by MIOX overexpression, while ameliorated by MIOX knockdown. Likewise, cisplatin-treated CD1 mice exhibited tubular damage and derangement of renal physiological parameters, which were alleviated by ferrostatin-1, a ferroptosis inhibitor. To investigate the relevance of MIOX to ferroptosis, WT mice, MIOX-overexpressing transgenic (MIOX-Tg) mice, and MIOX-KO mice were subjected to cisplatin treatment. In comparison with cisplatin-treated WT mice, cisplatin-treated MIOX-Tg mice had more severe renal pathological changes and perturbations in ferroptosis metabolic sensors, which were minimal in cisplatin-treated MIOX-KO mice. In conclusion, these findings indicate that ferroptosis, an integral process in the pathogenesis of cisplatin-induced AKI, is modulated by the expression profile of MIOX.
Keywords: Apoptosis; Nephrology.
Conflict of interest statement
Figures





References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
Miscellaneous

