First-line Immuno-Oncology Combination Therapies in Metastatic Renal-cell Carcinoma: Results from the International Metastatic Renal-cell Carcinoma Database Consortium
- PMID: 31445844
- PMCID: PMC6858928
- DOI: 10.1016/j.eururo.2019.07.048
First-line Immuno-Oncology Combination Therapies in Metastatic Renal-cell Carcinoma: Results from the International Metastatic Renal-cell Carcinoma Database Consortium
Abstract
Background: In metastatic renal-cell carcinoma (mRCC), recent data have shown efficacy of first-line ipilimumab and nivolumab (ipi-nivo) as well as immuno-oncology (IO)/vascular endothelial growth factor (VEGF) inhibitor combinations. Comparative data between these strategies are limited.
Objective: To compare the efficacy of ipi-nivo versus IO-VEGF (IOVE) combinations in mRCC, and describe practice patterns and effectiveness of second-line therapies.
Design, setting, and participants: Using the International Metastatic Renal-cell Carcinoma Database Consortium (IMDC) dataset, patients treated with any first-line IOVE combination were compared with those treated with ipi-nivo.
Intervention: All patients received first-line IO combination therapies.
Outcome measurements and statistical analysis: First- and second-line response rates, time to treatment failure (TTF), time to next treatment (TNT), and overall survival (OS) were analysed. Hazard ratios were adjusted for IMDC risk factors.
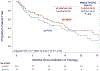
Results and limitations: In total, 113 patients received IOVE combinations and 75 received ipi-nivo. For IOVE combinations versus ipi-nivo, first-line response rates were 33% versus 40% (between-group difference 7%, 95% confidence interval [CI] -8% to 22%, p = 0.4), TTF was 14.3 versus 10.2 mo (p = 0.2), TNT was 19.7 versus 17.9 mo (p = 0.4), and median OS was immature but not statistically different (p = 0.17). Adjusted hazard ratios for TTF, TNT, and OS were 0.71 (95% CI 0.46-1.12, p = 0.14), 0.65 (95% CI 0.38-1.11, p = 0.11), and 1.74 (95% CI 0.82-3.68, p = 0.14), respectively. Sixty-four (34%) patients received second-line treatment. In patients receiving subsequent VEGF-based therapy, second-line response rates were lower in the IOVE cohort than in the ipi-nivo cohort (15% vs 45%; between-group difference 30%, 95% CI 3-57%, p = 0.04; n = 40), though second-line TTF was not significantly different (3.7 vs 5.4 mo; p = 0.4; n = 55). Limitations include the study's retrospective design and sample size.
Conclusions: There were no significant differences in first-line outcomes between IOVE combinations and ipi-nivo. Most patients received VEGF-based therapy in the second line. In this group, second-line response rate was greater in patients who received ipi-nivo initially.
Patient summary: There were no significant differences in key first-line outcomes for patients with metastatic renal-cell carcinoma receiving immuno-oncology/vascular endothelial growth factor inhibitor combinations versus ipilimumab and nivolumab.
Keywords: Checkpoint inhibitors; Combinations; First line; Immuno-oncology; International Metastatic Renal-cell Carcinoma Database Consortium; Metastatic; Programmed-Death 1 inhibitor; Renal-cell carcinoma; Survival.
Copyright © 2019 European Association of Urology. Published by Elsevier B.V. All rights reserved.
Figures


Comment in
-
Retrospective comparison of the different immune combinations in metastatic renal cell carcinoma.Ann Transl Med. 2020 Jul;8(13):839. doi: 10.21037/atm.2020.02.179. Ann Transl Med. 2020. PMID: 32793684 Free PMC article. No abstract available.
References
-
- Heng DY, Xie W, Regan MM, Warren MA, Golshayan AR, Sahi C, et al. Prognostic factors for overall survival in patients with metastatic renal cell carcinoma treated with vascular endothelial growth factor-targeted agents: results from a large, multicenter study. J Clin Oncol 2009;27:5794–9. - PubMed
-
- Lalani AA, McGregor BA, Albiges L, Choueiri TK, Motzer R, Powles T, et al. Systemic treatment of metastatic clear cell renal cell carcinoma in 2018: current paradigms, use of immunotherapy, and future directions. Eur Urol 2019;75:100–10. - PubMed
-
- Rini BI, Powles T, Atkins MB, Escudier B, McDermott DF, Suarez C, et al. Atezolizumab plus bevacizumab versus sunitinib in patients with previously untreated metastatic renal cell carcinoma (IMmotion151): a multicentre, open-label, phase 3, randomised controlled trial. Lancet 2019;393:2404–15. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

