Gold nanoparticles-loaded hydroxyapatite composites guide osteogenic differentiation of human mesenchymal stem cells through Wnt/β-catenin signaling pathway
- PMID: 31447557
- PMCID: PMC6683960
- DOI: 10.2147/IJN.S213889
Gold nanoparticles-loaded hydroxyapatite composites guide osteogenic differentiation of human mesenchymal stem cells through Wnt/β-catenin signaling pathway
Abstract
Background: Precise control and induction of the differentiation of stem cells to osteoblasts by artificial biomaterials are a promising strategy for rapid bone regeneration and reconstruction.
Purpose: In this study, gold nanoparticles (AuNPs)-loaded hydroxyapatite (HA-Au) nanocomposites were designed to guide the osteogenic differentiation of human bone marrow-derived mesenchymal stem cells (hMSCs) through the synergistic effects of both AuNPs and HA.
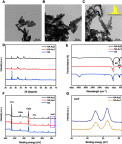
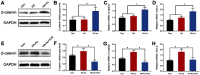
Materials and methods: The HA-Au nanoparticles were synthesized and characterized by several analytical techniques. Cell viability and proliferation of hMSCs were characterized by CCK-8 test. Cellular uptake of nanoparticles was observed by transmission electron microscope. For the evaluation of osteogenic differentiation, alkaline phosphatase (ALP) activity and staining, Alizarin red staining, and a quantitative real-time polymerase chain reaction (RT-PCR) analysis were performed. In order to examine specific signaling pathways, RT-PCR and Western blotting assay were performed.
Results: The results confirmed the successful synthesis of HA-Au nanocomposites. The HA-Au nanoparticles showed good cytocompatibility and internalized into hMSCs at the studied concentrations. The increased level of ALP production, deposition of calcium mineralization, as well as the expression of typical osteogenic genes, indicated the enhancement of osteogenic differentiation of hMSCs. Moreover, the incorporation of Au could activate the Wnt/β-catenin signaling pathway, which seemed to be the molecular mechanism underlying the osteoinductive capability of HA-Au nanoparticles.
Conclusion: The HA-Au nanoparticles exerted a synergistic effect on accelerating osteogenic differentiation of hMSCs, suggesting they may be potential candidates for bone repair and regeneration.
Keywords: bone regeneration; gold nanoparticles; hydroxyapatite; nanocomposites; osteoblast differentiation.
Conflict of interest statement
The authors report no conflicts of interest in this work.
Figures







References
-
- Naveena N, Venugopal J, Rajeswari R, et al. Biomimetic composites and stem cells interaction for bone and cartilage tissue regeneration. J Mater Chem. 2012;22(12):5239–5253. doi:10.1039/c1jm14401d - DOI
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

