Project management lessons learned from the multicentre CYCLE pilot randomized controlled trial
- PMID: 31455384
- PMCID: PMC6712681
- DOI: 10.1186/s13063-019-3634-7
Project management lessons learned from the multicentre CYCLE pilot randomized controlled trial
Erratum in
-
Correction to: Project management lessons learned from the multicentre CYCLE pilot randomized controlled trial.Trials. 2019 Oct 25;20(1):606. doi: 10.1186/s13063-019-3716-6. Trials. 2019. PMID: 31653225 Free PMC article.
Abstract
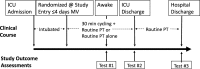
Background: Clinical trials management can be studied using project management theory. The CYCLE pilot randomized controlled trial (RCT) was conducted to determine the feasibility of a future rehabilitation trial of early in-bed cycling in the intensive care unit (ICU). In-bed cycling is a novel intervention, not typically available in ICUs. Implementation of this intervention requires personnel with specialized clinical expertise caring for critically ill patients and use of the in-bed cycle. Our objective was to describe the implementation and conduct of our pilot RCT using a project management approach.
Methods: We retrospectively reviewed activities, timelines, and personnel involved in the trial. We organized activities into four project management phases: initiation, planning, execution, and monitoring and controlling. Data sources included Methods Centre documents used for trial coordination and conduct, and the trial data set. We report descriptive statistics as counts and proportions and also medians and quartiles, and we summarize the lessons learned.
Results: Seven ICUs in Canada participated in the trial. Time from research ethics board and contracts submission to first enrolment was a median (first quartile, third quartile) of 185 (146, 209) and 162 (114, 181) days, respectively. We trained 128 personnel on the CYCLE pilot RCT protocol, and 80 (63%) completed trial-related activities. Four sites required additional training after start-up due to staff turnover and leaves of absence. Over 15 months, we screened 864 patients: 256 were eligible and 66 were enrolled. Despite an 85% consent rate, 74% (190/256) of eligible patients were not randomized, largely (80% [152/190]) due to physiotherapist availability. Thirteen percent of recruitment weeks were lost due to physiotherapist staffing shortages. We highlight five key lessons learned: (1) prepare and anticipate site needs; (2) communicate regularly; (3) proactively analyse and act on process measure data; (4) develop contingency plans; (5) express appreciation to participating sites.
Conclusions: Our analysis highlights the scope of relevant activities, rigorous training and monitoring, number and types of required personnel, and time required to conduct a multicentre ICU rehabilitation intervention trial. Our lessons learned can help others interested in implementing complex intervention trials, such as rehabilitation.
Trial registration: ClinicalTrials.gov, NCT02377830 . Registered prospectively on 4 March 2015.
Keywords: Critical care; Project management; Randomized controlled trial; Rehabilitation; Trial management.
Conflict of interest statement
Restorative Therapies (Baltimore, MD, USA) provided two RT-300 supine cycle ergometers for Toronto General Hospital and London Health Sciences sites for this research. The equipment manufacturer had no role in the design, execution, analysis, or interpretation of this study or in the decision to submit results.
Figures


References
-
- Chan Y. Randomised controlled trials (RCTs) — Essentials. Singap Med J. 2003;44(2):60–63. - PubMed
-
- Campbell MK, Snowdon C, Francis D, Elbourne D, McDonald AM, Knight R, et al. Recruitment to randomised trials: strategies for trial enrollment and participation study. The STEPS study. Health Technol Assess. 2007;11(48). - PubMed
MeSH terms
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

