Efficient Quantum Dot Light-Emitting Diodes Based on Trioctylphosphine Oxide-Passivated Organometallic Halide Perovskites
- PMID: 31460003
- PMCID: PMC6648744
- DOI: 10.1021/acsomega.9b00464
Efficient Quantum Dot Light-Emitting Diodes Based on Trioctylphosphine Oxide-Passivated Organometallic Halide Perovskites
Abstract
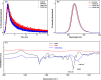
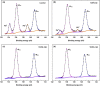
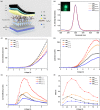
Metal halide perovskite quantum dots (QDs) have attracted significant research interest in the next-generation display and solid illumination fields due to their excellent optical properties of high photoluminescence quantum efficiency, high color purity, obvious quantum confinement effect, and large exciton binding energy. A large amount of surface defects and nonradiative recombination induced by these defects are considered as major problems to be resolved urgently for practical applications of perovskite QDs in high-efficiency light-emitting diodes (LEDs). Herein, we report an efficient passivation of green perovskite QD CH3NH3PbBr3 with trioctylphosphine oxide (TOPO). By simply adding the appropriate amount of TOPO into the nonpolar toluene solvent to synthesize CH3NH3PbBr3 QDs, the surface defects of these as-synthesized perovskite QDs are obviously reduced, along with an increased photoluminescence lifetime and suppressed nonradiative recombination. Further investigation indicates that electronegative oxygen from TOPO (Lewis base) bonds with uncoordinated Pb2+ ions and labile lead atoms in perovskite. With TOPO passivation, the green perovskite QD LEDs based on CH3NH3PbBr3 show significant performance improvement factors of 93.5, 161.1, and 168.9% for luminance, current efficiency, and external quantum efficiency, respectively, reaching values of 1635 cd m-2, 5.51 cd A-1, and 1.64% in the eventual optimized devices. Furthermore, the presence of TOPO dramatically improves stabilities of CH3NH3PbBr3 QDs and related devices. Our work provides a robust platform for the fabrication of low-defect-density perovskite QDs and efficient, stable perovskite QD LEDs.
Conflict of interest statement
The authors declare no competing financial interest.
Figures


Similar articles
-
Novel Lewis Base Cyclam Self-Passivation of Perovskites without an Anti-Solvent Process for Efficient Light-Emitting Diodes.ACS Appl Mater Interfaces. 2020 Mar 25;12(12):14224-14232. doi: 10.1021/acsami.0c02768. Epub 2020 Mar 16. ACS Appl Mater Interfaces. 2020. PMID: 32129073
-
CH3NH3PbBr3 Quantum Dot-Induced Nucleation for High Performance Perovskite Light-Emitting Solar Cells.ACS Appl Mater Interfaces. 2018 Jul 5;10(26):22320-22328. doi: 10.1021/acsami.8b06595. Epub 2018 Jun 22. ACS Appl Mater Interfaces. 2018. PMID: 29889489
-
Bilayer phosphine oxide modification toward efficient and large-area pure-blue perovskite quantum dot light-emitting diodes.Sci Bull (Beijing). 2023 Oct 30;68(20):2354-2361. doi: 10.1016/j.scib.2023.09.014. Epub 2023 Sep 12. Sci Bull (Beijing). 2023. PMID: 37730508
-
Versatile Defect Passivation Methods for Metal Halide Perovskite Materials and their Application to Light-Emitting Devices.Adv Mater. 2019 May;31(20):e1805244. doi: 10.1002/adma.201805244. Epub 2019 Jan 21. Adv Mater. 2019. PMID: 30663137 Review.
-
High Performance Metal Halide Perovskite Light-Emitting Diode: From Material Design to Device Optimization.Small. 2017 Dec;13(45). doi: 10.1002/smll.201701770. Epub 2017 Sep 29. Small. 2017. PMID: 28961367 Review.
Cited by
-
High-Performance Blue Quasi-2D Perovskite Light-Emitting Diodes via Balanced Carrier Confinement and Transfer.Nanomicro Lett. 2022 Feb 23;14(1):66. doi: 10.1007/s40820-022-00807-7. Nanomicro Lett. 2022. PMID: 35199224 Free PMC article.
-
Flexible Colloidal Light-Emitting Diodes of Self-Assembled Quantum Well Monolayers.Small. 2025 Aug;21(32):e2502314. doi: 10.1002/smll.202502314. Epub 2025 May 3. Small. 2025. PMID: 40317681 Free PMC article.
References
-
- Shi D.; Adinolfi V.; Comin R.; Yuan M.; Alarousu E.; Buin A.; Chen Y.; Hoogland S.; Rothenberger A.; Katsiev K.; Losovyj Y.; Zhang X.; Dowben P. A.; Mohammed O. F.; Sargent E. H.; Bakr O. M. Low trap-state density and long carrier diffusion in organolead trihalide perovskite single crystals. Science 2015, 347, 519–522. 10.1126/science.aaa2725. - DOI - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources

