A Sulfonamide Sialoside Analogue for Targeting Siglec-8 and -F on Immune Cells
- PMID: 31460762
- PMCID: PMC6861165
- DOI: 10.1021/jacs.9b05769
A Sulfonamide Sialoside Analogue for Targeting Siglec-8 and -F on Immune Cells
Abstract
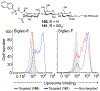
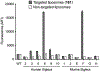
The Siglec family of cell surface receptors have emerged as attractive targets for cell-directed therapies due to their restricted expression on immune cells, endocytic properties, and ability to modulate receptor signaling. Human Siglec-8, for instance, has been identified as a therapeutic target for the treatment of eosinophil and mast cell disorders. A promising strategy to target Siglecs involves the use of liposomal nanoparticles with a multivalent display of Siglec ligands. A key challenge for this approach is the identification of a high affinity ligand for the target Siglec. Here, we report the development of a ligand of Siglec-8 and its closest murine functional orthologue Siglec-F that is capable of targeting liposomes to cells expressing Siglec-8 or -F. A glycan microarray library of synthetic 9-N-sulfonyl sialoside analogues was screened to identify potential lead compounds. The best ligand, 9-N-(2-naphthyl-sulfonyl)-Neu5Acα2-3-[6-O-sulfo]-Galβ1-4GlcNAc (6'-O-sulfo NSANeu5Ac) combined the lead 2-naphthyl sulfonyl C-9 substituent with the preferred sulfated scaffold. The ligand 6'-O-sulfo NSANeu5Ac was conjugated to lipids for display on liposomes to evaluate targeted delivery to cells. Targeted liposomes showed strong in vitro binding/uptake and selectivity to cells expressing Siglec-8 or -F and, when administered to mice, exhibit in vivo targeting to Siglec-F+ eosinophils.
Conflict of interest statement
Dr. Bochner receives remuneration for serving on the scientific advisory board of Allakos, Inc.; and owns stock in Allakos. He receives publication-related royalty payments from Elsevier and Up-ToDate®. He is a co-inventor on existing Siglec-8-related patents and thus may be entitled to a share of royalties received by Johns Hopkins University during development and potential sales of such products. Dr. Bochner is also a co-founder of Allakos, which makes him subject to certain restrictions under University policy. The terms of this arrangement are being managed by the Johns Hopkins University and Northwestern University in accordance with their conflict of interest policies.
All other authors declare no competing financial interests.
Figures

Similar articles
-
Mouse Siglec-F and human Siglec-8 are functionally convergent paralogs that are selectively expressed on eosinophils and recognize 6'-sulfo-sialyl Lewis X as a preferred glycan ligand.Glycobiology. 2005 Nov;15(11):1125-35. doi: 10.1093/glycob/cwi097. Epub 2005 Jun 22. Glycobiology. 2005. PMID: 15972893
-
Siglec-8 on human eosinophils and mast cells, and Siglec-F on murine eosinophils, are functionally related inhibitory receptors.Clin Exp Allergy. 2009 Mar;39(3):317-24. doi: 10.1111/j.1365-2222.2008.03173.x. Clin Exp Allergy. 2009. PMID: 19178537 Free PMC article. Review.
-
Probing sialic acid binding Ig-like lectins (siglecs) with sulfated oligosaccharides.Biochemistry (Mosc). 2006 May;71(5):496-504. doi: 10.1134/s0006297906050051. Biochemistry (Mosc). 2006. PMID: 16732727
-
On-chip synthesis and screening of a sialoside library yields a high affinity ligand for Siglec-7.ACS Chem Biol. 2013 Jul 19;8(7):1417-22. doi: 10.1021/cb400125w. Epub 2013 Apr 24. ACS Chem Biol. 2013. PMID: 23597400 Free PMC article.
-
Siglec-8 as a drugable target to treat eosinophil and mast cell-associated conditions.Pharmacol Ther. 2012 Sep;135(3):327-36. doi: 10.1016/j.pharmthera.2012.06.005. Epub 2012 Jun 27. Pharmacol Ther. 2012. PMID: 22749793 Free PMC article. Review.
Cited by
-
Emerging glyco-based strategies to steer immune responses.FEBS J. 2021 Aug;288(16):4746-4772. doi: 10.1111/febs.15830. Epub 2021 May 15. FEBS J. 2021. PMID: 33752265 Free PMC article. Review.
-
Recent progress in targeting the sialylated glycan-SIGLEC axis in cancer immunotherapy.Cancer Biol Med. 2023 May 3;20(5):369-84. doi: 10.20892/j.issn.2095-3941.2023.0046. Cancer Biol Med. 2023. PMID: 37133224 Free PMC article. Review.
-
Structures of the Inhibitory Receptor Siglec-8 in Complex with a High-Affinity Sialoside Analogue and a Therapeutic Antibody.JACS Au. 2022 Dec 23;3(1):204-215. doi: 10.1021/jacsau.2c00592. eCollection 2023 Jan 23. JACS Au. 2022. PMID: 36711084 Free PMC article.
-
Targeted delivery of oligonucleotides using multivalent protein-carbohydrate interactions.Chem Soc Rev. 2023 Feb 20;52(4):1273-1287. doi: 10.1039/d2cs00788f. Chem Soc Rev. 2023. PMID: 36723021 Free PMC article. Review.
-
Unraveling Molecular Recognition of Glycan Ligands by Siglec-9 via NMR Spectroscopy and Molecular Dynamics Modeling.ACS Chem Biol. 2024 Feb 16;19(2):483-496. doi: 10.1021/acschembio.3c00664. Epub 2024 Feb 7. ACS Chem Biol. 2024. PMID: 38321945 Free PMC article.
References
-
- Crocker PR; Paulson JC; Varki A, Siglecs and their roles in the immune system. Nat Rev Immunol 2007, 7 (4), 255–266. - PubMed
-
- Adams OJ; Stanczak MA; von Gunten S; Laubli H, Targeting sialic acid-Siglec interactions to reverse immune suppression in cancer. Glycobiology 2018, 28 (9), 640–647. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

