Pancreatic cancer-associated inflammation drives dynamic regulation of p35 and Ebi3
- PMID: 31472403
- PMCID: PMC6876557
- DOI: 10.1016/j.cyto.2019.154817
Pancreatic cancer-associated inflammation drives dynamic regulation of p35 and Ebi3
Abstract
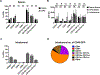
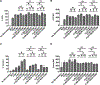
B cells are important modulators of immune responses both in autoimmunity and cancer. We have previously shown that B regulatory (Breg) cells promote pancreatic cancer via production of IL35, a heterodimeric cytokine comprised of the subunits p35 (Il12a) and Ebi3. However, it is not known how production of IL35 is regulated in vivo in the context of cancer-associated inflammation. To begin addressing this question, we have generated a knock-in mouse model, Il12aGFP, where an IRES-emGFP gene was inserted within the 3' UTR of the Il12a locus. EmGFP signal in B cells from the Il12aGFP mice correlated with expression of p35 mRNA and protein. Using this model, we observed that in addition to Bregs, expression of GFP (p35) is upregulated in several other B cell subtypes in response to cancer. We assessed the expression of the other IL35 subunit, Ebi3, using a published tdTomato reporter model. We determined that Ebi3 expression was more tightly regulated in vivo and in vitro, suggesting that stimuli affecting Ebi3 upregulation are more likely to result in production of full IL35 heterodimer. We were also able to detect GFP and Tomato signal in myeloid & T cell lineages suggesting that these reporter models could also be used for tracking IL12-, IL27- and IL35-producing cells. Furthermore, using primary B cells isolated from reporter mice, we identified BCR, CD40 and TLR pathways as potential drivers of IL35 expression. These findings highlight the importance of pancreatic cancer-associated inflammatory processes as drivers of cytokine expression and provide a tool to dissect both disease-associated regulation of IL12- and IL35-competent lineage cells as well as establish assays for pharmacological targeting of individual subunits of heterodimeric IL12 family cytokines.
Keywords: IL35 cytokine; Pancreatic cancer; Regulatory B cell; Reporter model.
Copyright © 2019 Elsevier Ltd. All rights reserved.
Conflict of interest statement
COMPETING FINANCIAL INTERESTS
D.A.A.V. has submitted patents covering IL-35 that are pending and are entitled to a share in net income generated from licensing of these patent rights for commercial development. D.O.C. is employed by, has equity ownership in and serves on the board of directors of TransViragen, the company which has been contracted by UNC-Chapel Hill to manage its Animal Models Core Facility.
Declaration of interests
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Figures



References
-
- Shen P, Roch T, Lampropoulou V, O’Connor RA, Stervbo U, Hilgenberg E, Ries S, Dang VD, Jaimes Y, Daridon C, Li R, Jouneau L, Boudinot P, Wilantri S, Sakwa I, Miyazaki Y, Leech MD, McPherson RC, Wirtz S, Neurath M, Hoehlig K, Meinl E, Grutzkau A, Grun JR, Horn K, Kuhl AA, Dorner T, Bar-Or A, Kaufmann SHE, Anderton SM, Fillatreau S. IL-35-producing B cells are critical regulators of immunity during autoimmune and infectious diseases. Nature 2014;507(7492):366–70. Epub 2014/02/28. doi: 10.1038/nature12979. - DOI - PMC - PubMed
-
- Dambuza IM, He C, Choi JK, Yu CR, Wang R, Mattapallil MJ, Wingfield PT, Caspi RR, Egwuagu CE. IL-12p35 induces expansion of IL-10 and IL-35-expressing regulatory B cells and ameliorates autoimmune disease. Nat Commun 2017;8(1):719 Epub 2017/09/30. doi: 10.1038/s41467-017-00838-4. - DOI - PMC - PubMed
-
- Sun T, Zhang D, Yang Y, Zhang X, Lv C, Fu R, Lv M, Liu W, Chen Y, Liu W, Huang Y, Xue F, Liu X, Zhang L, Li H, Yang R. Interleukin 35 may contribute to the loss of immunological self-tolerance in patients with primary immune thrombocytopenia. Br J Haematol 2015;169(2):278–85. Epub 2015/02/03. doi: 10.1111/bjh.13292. - DOI - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Research Materials

