Cardiac troponin I autoantibody induces myocardial dysfunction by PTEN signaling activation
- PMID: 31474552
- PMCID: PMC6796505
- DOI: 10.1016/j.ebiom.2019.08.045
Cardiac troponin I autoantibody induces myocardial dysfunction by PTEN signaling activation
Abstract
Background: The objective of the current study was to study the molecular mechanism(s) underlying cardiac troponin I autoantibody (cTnIAAb) binding to cardiomyocyte and resultant myocardial damage/dysfunction.
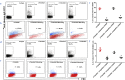
Methods: cTnIAAb was purified from serum of 10 acute myocardial infarction (AMI) patients with left ventricular remodeling. Recombinant human cTnI was used to generate three mouse-derived monoclonal anti-cTnI antibodies (cTnImAb1, cTnImAb2, and cTnImAb3). The target proteins in cardiac myocyte membrane bound to cTnImAb and effect of cTnIAAb and cTnImAb on apoptosis and myocardial function were determined.
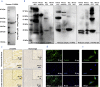
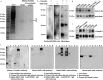
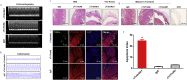
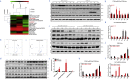
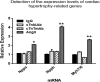
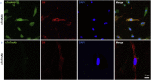
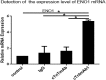
Findings: We found that cTnIAAb/cTnImAb1 directly bound to the cardiomyocyte membraneα-Enolase (ENO1) and triggered cell apoptosis via increased expression of ENO1 and Bax, decreased expression of Bcl2, subsequently activating Caspase8, Caspase 3, phosphatase and tensin homolog (PTEN) while inhibiting Akt activity. This cTnIAAb-ENO1-PTEN-Akt signaling axis contributed to increased myocardial apoptosis, myocardial collagen deposition, and impaired systolic dysfunction.
Interpretation: Results obtained in this study indicate that cTnIAAb is involved in the process of ventricular remodeling after myocardial injury. FUND: The National Natural Science Foundation of China (Grant#: 81260026).
Keywords: Acute myocardial infarction; Cardiac troponin I autoantibody; Phosphatase and tensin homolog; Ventricular remodeling; α-Enolase.
Copyright © 2019. Published by Elsevier B.V.
Figures






References
-
- Tang G., Wu Y., Zhao W. Multiple immunoassay systems are negatively interfered by circulating cardiac troponin I autoantibodies. Clin Exp Med. 2012;12:47–53. - PubMed
-
- Shmilovich H., Danon A., Binah O. Autoantibodies to cardiac troponin I in patients with idiopathic dilated and ischemic cardiomyopathy. Int J Cardiol. 2007;117:198–203. - PubMed
-
- Eriksson S., Hellman J., Pettersson K. Autoantibodies against cardiac troponins. N Engl J Med. 2005;352:98–100. - PubMed
-
- Pettersson K., Eriksson S., Wittfooth S. Autoantibodies to cardiac troponin associate with higher initial concentrations and longer release of troponin I in acute coronary syndrome patients. Clin Chem. 2009;55:938–945. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
Miscellaneous

