Low Frequency Systemic Hemodynamic "Noise" in Resting State BOLD fMRI: Characteristics, Causes, Implications, Mitigation Strategies, and Applications
- PMID: 31474815
- PMCID: PMC6702789
- DOI: 10.3389/fnins.2019.00787
Low Frequency Systemic Hemodynamic "Noise" in Resting State BOLD fMRI: Characteristics, Causes, Implications, Mitigation Strategies, and Applications
Abstract
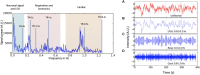
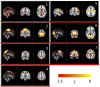
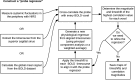
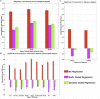
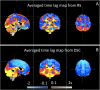
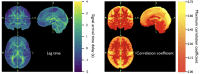
Advances in functional magnetic resonance imaging (fMRI) acquisition have improved signal to noise to the point where the physiology of the subject is the dominant noise source in resting state fMRI data (rsfMRI). Among these systemic, non-neuronal physiological signals, respiration and to some degree cardiac fluctuations can be removed through modeling, or in the case of newer, faster acquisitions such as simultaneous multislice acquisition, simple spectral filtering. However, significant low frequency physiological oscillation (∼0.01-0.15 Hz) remains in the signal. This is problematic, as it is the precise frequency band occupied by the neuronally modulated hemodynamic responses used to study brain connectivity, precluding its removal by spectral filtering. The source of this signal, and its method of production and propagation in the body, have not been conclusively determined. Here, we summarize the defining characteristics of the systemic low frequency noise signal, and review some current theories about the signal source and the evidence supporting them. The strength and distribution of the systemic LFO signal make characterizing and removing it essential for accurate quantification, especially for resting state connectivity, when no stimulation can be compared with the signal. Widespread correlated non-neuronal signals obscure and distort the more localized patterns of neuronal correlations between interacting brain regions; they may even cause apparent connectivity between regions with no neuronal interaction. Here, we discuss a simple method we have developed to parse the global, moving, blood-borne signal from the stationary, neuronal connectivity signals, substantially reducing the negative correlations that result from global signal regression. Finally, we will discuss some of the uses to which the moving systemic low frequency oscillation can be put if we consider it a "signal" carrying information, rather than simply "noise" complicating the interpretation of resting state connectivity. Properly utilizing this signal may offer insights into subtle hemodynamic alterations that can be used as early indicators of circulatory dysfunction in a number of neuropsychiatric conditions, such as prodromal stroke, moyamoya, and Alzheimer's disease.
Keywords: cerebrovascular reactivity; denoising; low frequency oscillation; noise modeling; physiological noise; physiological noise modeling; vascular mapping.
Figures


Similar articles
-
Estimating and mitigating the effects of systemic low frequency oscillations (sLFO) on resting state networks in awake non-human primates using time lag dependent methodology.Front Neuroimaging. 2023 Jan 19;1:1031991. doi: 10.3389/fnimg.2022.1031991. eCollection 2022. Front Neuroimaging. 2023. PMID: 37555145 Free PMC article.
-
Correcting for Blood Arrival Time in Global Mean Regression Enhances Functional Connectivity Analysis of Resting State fMRI-BOLD Signals.Front Hum Neurosci. 2016 Jun 28;10:311. doi: 10.3389/fnhum.2016.00311. eCollection 2016. Front Hum Neurosci. 2016. PMID: 27445751 Free PMC article.
-
A kernel machine-based fMRI physiological noise removal method.Magn Reson Imaging. 2014 Feb;32(2):150-62. doi: 10.1016/j.mri.2013.10.008. Epub 2013 Oct 19. Magn Reson Imaging. 2014. PMID: 24321306 Free PMC article.
-
Functional MRI and resting state connectivity in white matter - a mini-review.Magn Reson Imaging. 2019 Nov;63:1-11. doi: 10.1016/j.mri.2019.07.017. Epub 2019 Jul 31. Magn Reson Imaging. 2019. PMID: 31376477 Free PMC article. Review.
-
Biophysical and neural basis of resting state functional connectivity: Evidence from non-human primates.Magn Reson Imaging. 2017 Jun;39:71-81. doi: 10.1016/j.mri.2017.01.020. Epub 2017 Feb 2. Magn Reson Imaging. 2017. PMID: 28161319 Free PMC article. Review.
Cited by
-
An orderly sequence of autonomic and neural events at transient arousal changes.Neuroimage. 2022 Dec 1;264:119720. doi: 10.1016/j.neuroimage.2022.119720. Epub 2022 Nov 2. Neuroimage. 2022. PMID: 36332366 Free PMC article.
-
Test-retest reliability of dynamic functional connectivity in naturalistic paradigm functional magnetic resonance imaging.Hum Brain Mapp. 2022 Mar;43(4):1463-1476. doi: 10.1002/hbm.25736. Epub 2021 Dec 6. Hum Brain Mapp. 2022. PMID: 34870361 Free PMC article.
-
The diagnostic potential of resting state functional MRI: Statistical concerns.Neuroimage. 2025 Aug 15;317:121334. doi: 10.1016/j.neuroimage.2025.121334. Epub 2025 Jun 17. Neuroimage. 2025. PMID: 40554035 Free PMC article. Review.
-
Using carpet plots to analyze transit times of low frequency oscillations in resting state fMRI.Sci Rep. 2021 Mar 26;11(1):7011. doi: 10.1038/s41598-021-86402-z. Sci Rep. 2021. PMID: 33772060 Free PMC article.
-
Cerebrovascular stiffness and flow dynamics in the presence of amyloid and tau biomarkers.Alzheimers Dement (Amst). 2021 Dec 31;13(1):e12253. doi: 10.1002/dad2.12253. eCollection 2021. Alzheimers Dement (Amst). 2021. PMID: 35005194 Free PMC article.
References
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources

