The Methanol Extract of Allium cepa L. Protects Inflammatory Markers in LPS-Induced BV-2 Microglial Cells and Upregulates the Antiapoptotic Gene and Antioxidant Enzymes in N27-A Cells
- PMID: 31480531
- PMCID: PMC6770444
- DOI: 10.3390/antiox8090348
The Methanol Extract of Allium cepa L. Protects Inflammatory Markers in LPS-Induced BV-2 Microglial Cells and Upregulates the Antiapoptotic Gene and Antioxidant Enzymes in N27-A Cells
Abstract
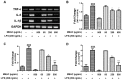
Neuroinflammation, apoptosis, and oxidative stress are connected to the pathogenesis of neurodegenerative diseases (NDDs). Targeting these three factors, the intervention of neuroprotective agents may have great potential in the treatment of NDDs. In the current study, the anti-inflammatory effects of the methanol extract of Allium cepa (MEAC) in lipopolysaccharide (LPS)-induced BV-2 microglial cells were investigated. MEAC has been studied in regard to the regulation of the antiapoptotic gene (Bcl-2) and various antioxidant enzyme (HO-1, NQO-1, and catalase) expressions in N27-A cells. Additionally, the protective action of MEAC has also been studied against MPP+-induced death in N27-A cells. The results suggest that MEAC is significantly protected from NO release and increase iNOS expression at the mRNA and protein levels in LPS-stimulated BV-2 microglial cells. MEAC treatment also protects COX-2 expression at the mRNA and protein levels. Furthermore, MEAC treatment prevents LPS-stimulated increases of proinflammatory cytokines, including TNF-α, IL-6, and IL-1β. In N27-A cells, MEAC treatment significantly upregulates antiapoptotic gene (Bcl-2) and antioxidant enzyme (HO-1, NQO1, and catalase) expressions. Moreover, MEAC treatment protects against MPP+-induced death in N27-A cells. To conclude, A cepa extract takes protective action against LPS and MPP+, and upregulates the antioxidant enzymes that could potentially be used in the therapy of NDDs.
Keywords: Allium cepa; Bcl-2; antioxidant enzymes; inflammatory markers; neurodegenerative diseases; protection.
Conflict of interest statement
The authors declare that they have no conflicts of interest.
Figures





References
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

