Lactobacillus reuteri DSM 17938 feeding of healthy newborn mice regulates immune responses while modulating gut microbiota and boosting beneficial metabolites
- PMID: 31482733
- PMCID: PMC6962498
- DOI: 10.1152/ajpgi.00107.2019
Lactobacillus reuteri DSM 17938 feeding of healthy newborn mice regulates immune responses while modulating gut microbiota and boosting beneficial metabolites
Abstract
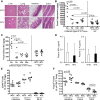
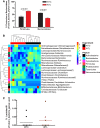
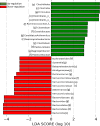
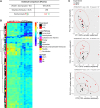
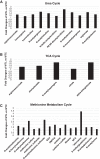
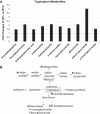
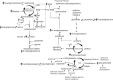
Early administration of Lactobacillus reuteri DSM 17938 (LR) prevents necrotizing enterocolitis and inhibits regulatory T-cell (Treg)-deficiency-associated autoimmunity in mice. In humans, LR reduces crying time in breastfed infants with colic, modifies severity in infants with acute diarrheal illnesses, and improves pain in children with functional bowel disorders. In healthy breastfed newborns with evolving microbial colonization, it is unclear if early administration of LR can modulate gut microbiota and their metabolites in such a way as to promote homeostasis. We gavaged LR (107 colony-forming units/day, daily) to C57BL/6J mice at age of day 8 for 2 wk. Both male and female mice were investigated in these experiments. We found that feeding LR did not affect clinical phenotype or inflammatory biomarkers in plasma and stool, but LR increased the proportion of Foxp3+ regulatory T cells (Tregs) in the intestine. LR also increased bacterial diversity and the relative abundance of p_Firmicutes, f_Lachnospiraceae, f_Ruminococcaceae, and genera Clostridium and Candidatus arthromitus, while decreasing the relative abundance of p_Bacteriodetes, f_Bacteroidaceae, f_Verrucomicrobiaceae, and genera Bacteroides, Ruminococcus, Akkermansia, and Sutterella. Finally, LR exerted a major impact on the plasma metabolome, upregulating amino acid metabolites formed via the urea, tricarboxylic acid, and methionine cycles and increasing tryptophan metabolism. In conclusion, early oral administration of LR to healthy breastfed mice led to microbial and metabolic changes which could be beneficial to general health.NEW & NOTEWORTHY Oral administration of Lactobacillus reuteri DSM 17938 (LR) to healthy breastfed mice promotes intestinal immune tolerance and is linked to proliferation of beneficial gut microbiota. LR upregulates plasma metabolites that are involved in the urea cycle, the TCA cycle, methionine methylation, and the polyamine pathway. Herein, we show that LR given to newborn mice specifically increases levels of tryptophan metabolites and the purine nucleoside adenosine that are known to enhance tolerance to inflammatory stimuli.
Keywords: Bacteroidetes; Firmicutes; biomarker; healthy; intestine; lactobacillus reuteri; metabolites; microbiota; newborn; probiotic.
Conflict of interest statement
No conflicts of interest, financial or otherwise, are declared by the authors.
Figures




References
-
- Biddle A, Stewart L, Blanchard J, Leschine S. Untangling the genetic basis of fibrolytic specialization by Lachnospiraceae and Ruminococcaceae in diverse gut communities. Diversity (Basel) 5: 627–640, 2013. doi: 10.3390/d5030627. - DOI
-
- Bolotin A, de Wouters T, Schnupf P, Bouchier C, Loux V, Rhimi M, Jamet A, Dervyn R, Boudebbouze S, Blottière HM, Sorokin A, Snel J, Cerf-Bensussan N, Gaboriau-Routhiau V, van de Guchte M, Maguin E. Genome sequence of “Candidatus Arthromitus” sp. Strain SFB-mouse-NL, a commensal bacterium with a key role in postnatal maturation of gut immune functions. Genome Announc 2: e00705-14, 2014. doi: 10.1128/genomeA.00705-14. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

