Quantifying the impact of tissue metabolism on solute transport in feto-placental microvascular networks
- PMID: 31485311
- PMCID: PMC6710657
- DOI: 10.1098/rsfs.2019.0021
Quantifying the impact of tissue metabolism on solute transport in feto-placental microvascular networks
Abstract
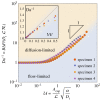
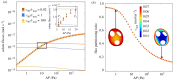
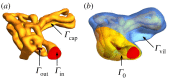
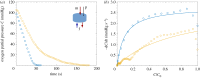
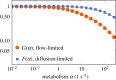
The primary exchange units in the human placenta are terminal villi, in which fetal capillary networks are surrounded by a thin layer of villous tissue, separating fetal from maternal blood. To understand how the complex spatial structure of villi influences their function, we use an image-based theoretical model to study the effect of tissue metabolism on the transport of solutes from maternal blood into the fetal circulation. For solute that is taken up under first-order kinetics, we show that the transition between flow-limited and diffusion-limited transport depends on two new dimensionless parameters defined in terms of key geometric quantities, with strong solute uptake promoting flow-limited transport conditions. We present a simple algebraic approximation for solute uptake rate as a function of flow conditions, metabolic rate and villous geometry. For oxygen, accounting for nonlinear kinetics using physiological parameter values, our model predicts that villous metabolism does not significantly impact oxygen transfer to fetal blood, although the partitioning of fluxes between the villous tissue and the capillary network depends strongly on the flow regime.
Keywords: human placenta; metabolism; microcirculation; solute transport.
Conflict of interest statement
The authors declare that they have no competing interests.
Figures


References
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources

