Long-Term Results of Cultured Limbal Stem Cell Versus Limbal Tissue Transplantation in Stage III Limbal Deficiency
- PMID: 31486585
- PMCID: PMC6877763
- DOI: 10.1002/sctm.19-0021
Long-Term Results of Cultured Limbal Stem Cell Versus Limbal Tissue Transplantation in Stage III Limbal Deficiency
Abstract
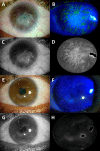
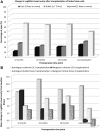
We aimed to evaluate efficiency and safety of transplantation of limbal stem cells (LSC) cultured on human amniotic membrane with no feeders and to compare cultured LSC with limbal tissue transplantation. Thirty eyes with stage III LSC deficiency were treated with autologous (autoLSC) or allogeneic (alloLSC) cultured LSC transplantation (prospective phase II clinical trial; average follow-up time, 72 months) or autologous (autoLT) or allogeneic (alloLT) limbal tissue transplantation (retrospective control group; average follow-up time, 132 months) between 1993 and 2014. The 5-year graft survival defined by absence of recurrence of the clinical signs of limbal deficiency was 71% for autoLSC, 0% for alloLSC, 75% for autoLT, and 33% for alloLT. Visual acuity improved by 9.2 lines for autoLSC and 3.3 lines for autoLT. It decreased by 0.7 lines for alloLSC and 1.9 lines for alloLT. Adverse events were recorded in 1/7 autoLSC, 7/7 alloLSC, 6/8 autoLT, and 8/8 alloLT patients. Corneal epithelial defect was the only adverse event recorded after autoLSC, whereas severe sight-threatening adverse events were recorded in the remaining three groups. Compared with failed grafts, successful grafts featured greater decrease in fluorescein staining, greater superficial vascularization-free corneal area, lower variability of the corneal epithelial thickness, and higher corneal epithelial basal cell density. Autologous cultured LSC transplantation was associated with high long-term survival and dramatic improvement in vision and was very safe. Autologous limbal tissue transplantation resulted in similar efficiency but was less safe. Cadaver allogeneic grafts resulted in low long-term success rate and high prevalence of serious adverse events. Stem Cells Translational Medicine 2019;8:1230&1241.
Keywords: Adverse events; Cornea; Limbal stem cell; Survival; Transplantation; Visual acuity.
© 2019 The Authors. Stem Cells Translational Medicine published by Wiley Periodicals, Inc. on behalf of AlphaMed Press.
Conflict of interest statement
V.M.B. declared consultant/advisory role with Chiesi (Parma, Italy; consulting), Dompe (Milan, Italy; consulting), and Optovue (Fremont, CA; consulting). The other authors indicated no potential conflicts of interest.
Figures


Similar articles
-
Clinical outcomes of penetrating keratoplasty after autologous cultivated limbal epithelial transplantation for ocular surface burns.Am J Ophthalmol. 2011 Dec;152(6):917-924.e1. doi: 10.1016/j.ajo.2011.05.019. Am J Ophthalmol. 2011. PMID: 21851920
-
Long-term effectiveness of autologous cultured limbal stem cell grafts in patients with limbal stem cell deficiency due to chemical burns.Clin Exp Ophthalmol. 2012 Apr;40(3):255-67. doi: 10.1111/j.1442-9071.2011.02609.x. Epub 2011 Aug 3. Clin Exp Ophthalmol. 2012. PMID: 21668791
-
Amniotic membrane transplantation for partial and total limbal stem cell deficiency secondary to chemical burn.Ophthalmology. 2003 Mar;110(3):466-73. doi: 10.1016/s0161-6420(02)01888-2. Ophthalmology. 2003. PMID: 12623806
-
Autologous limbal stem cell transplantation: a systematic review of clinical outcomes with different surgical techniques.Br J Ophthalmol. 2020 Feb;104(2):247-253. doi: 10.1136/bjophthalmol-2019-314081. Epub 2019 May 22. Br J Ophthalmol. 2020. PMID: 31118185
-
Midterm results of cultivated autologous and allogeneic limbal epithelial transplantation in limbal stem cell deficiency.Dev Ophthalmol. 2010;45:57-70. doi: 10.1159/000315020. Epub 2010 May 18. Dev Ophthalmol. 2010. PMID: 20502027 Review.
Cited by
-
Process development and safety evaluation of ABCB5+ limbal stem cells as advanced-therapy medicinal product to treat limbal stem cell deficiency.Stem Cell Res Ther. 2021 Mar 19;12(1):194. doi: 10.1186/s13287-021-02272-2. Stem Cell Res Ther. 2021. PMID: 33741066 Free PMC article.
-
Induction of Corneal Epithelial Differentiation of Induced Pluripotent and Orbital Fat-Derived Stem Cells Seeded on Decellularized Human Corneas.Stem Cell Rev Rep. 2022 Oct;18(7):2522-2534. doi: 10.1007/s12015-022-10356-6. Epub 2022 Mar 5. Stem Cell Rev Rep. 2022. PMID: 35247143
-
Long-term survival of cultivated oral mucosal epithelial cells in human cornea: generating cell sheets using an animal product-free culture protocol.Stem Cell Res Ther. 2021 Oct 7;12(1):524. doi: 10.1186/s13287-021-02564-7. Stem Cell Res Ther. 2021. PMID: 34620226 Free PMC article.
-
Niche regulation of limbal epithelial stem cells: HC-HA/PTX3 as surrogate matrix niche.Exp Eye Res. 2020 Oct;199:108181. doi: 10.1016/j.exer.2020.108181. Epub 2020 Aug 12. Exp Eye Res. 2020. PMID: 32795525 Free PMC article. Review.
-
Recent Advances in Stem Cell Therapy for Limbal Stem Cell Deficiency: A Narrative Review.Ophthalmol Ther. 2020 Dec;9(4):809-831. doi: 10.1007/s40123-020-00305-2. Epub 2020 Sep 24. Ophthalmol Ther. 2020. PMID: 32970311 Free PMC article. Review.
References
-
- Dua HS, Azuara‐Blanco A. Limbal stem cells of the corneal epithelium. Surv Ophthalmol 2000;44:415–425. - PubMed
-
- Pellegrini G, Traverso CE, Franzi AT et al. Long‐term restoration of damaged corneal surfaces with autologous cultivated corneal epithelium. Lancet 1997;349:990–993. - PubMed
-
- Tsai RJ, Li LM, Chen JK. Reconstruction of damaged corneas by transplantation of autologous limbal epithelial cells. N Engl J Med 2000;343:86–93. - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Medical

