MTA1-Dependent Anticancer Activity of Gnetin C in Prostate Cancer
- PMID: 31487842
- PMCID: PMC6770780
- DOI: 10.3390/nu11092096
MTA1-Dependent Anticancer Activity of Gnetin C in Prostate Cancer
Abstract
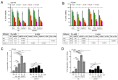
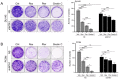
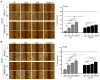
The overexpression of metastasis-associated protein 1 (MTA1) in prostate cancer (PCa) contributes to tumor aggressiveness and metastasis. We have reported the inhibition of MTA1 by resveratrol and its potent analog pterostilbene in vitro and in vivo. We have demonstrated that pterostilbene treatment blocks the progression of prostatic intraepithelial neoplasia and adenocarcinoma in mouse models by inhibiting MTA1 expression and signaling. In the current study, we investigated the MTA1 targeted anticancer effects of Gnetin C, a resveratrol dimer, in comparison with resveratrol and pterostilbene. Using DU145 and PC3M PCa cells, we found that Gnetin C downregulates MTA1 more potently than resveratrol and pterostilbene. Further, Gnetin C demonstrated significant MTA1-mediated inhibitory effect on cell viability, colony formation, and migration, while showing a more potent induction of cell death than resveratrol or pterostilbene. In addition, we identified Gnetin C-induced substantial ETS2 (erythroblastosis E26 transformation-specific 2) downregulation, which is not only MTA1-dependent, but is also independent of MTA1 as a possible mechanism for the superior anticancer efficacy of Gnetin C in PCa. Together, these findings underscore the importance of novel potent resveratrol dimer, Gnetin C, as a clinically promising agent for the future development of chemopreventive and possibly combinatorial therapeutic approaches in PCa.
Keywords: ETS2; Gnetin C; MTA1; prostate cancer; pterostilbene; resveratrol.
Conflict of interest statement
The authors declare no conflict of interests.
Figures




References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

