Creation of a long-acting rilpivirine prodrug nanoformulation
- PMID: 31491432
- PMCID: PMC6875695
- DOI: 10.1016/j.jconrel.2019.09.001
Creation of a long-acting rilpivirine prodrug nanoformulation
Abstract
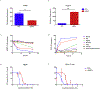
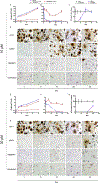
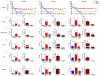
Antiretroviral therapy requires lifelong daily dosing to attain viral suppression, restore immune function, and improve quality of life. As a treatment alternative, long-acting (LA) antiretrovirals can sustain therapeutic drug concentrations in blood for prolonged time periods. The success of recent clinical trials for LA parenteral cabotegravir and rilpivirine highlight the emergence of these new therapeutic options. Further optimization can improve dosing frequency, lower injection volumes, and facilitate drug-tissue distributions. To this end, we report the synthesis of a library of RPV prodrugs designed to sustain drug plasma concentrations and improved tissue biodistribution. The lead prodrug M3RPV was nanoformulated into the stable LA injectable NM3RPV. NM3RPV treatment led to RPV plasma concentrations above the protein-adjusted 90% inhibitory concentration for 25 weeks with substantial tissue depots after a single intramuscular injection in BALB/cJ mice. NM3RPV elicited 13- and 26-fold increases in the RPV apparent half-life and mean residence time compared to native drug formulation. Taken together, proof-of-concept is provided that nanoformulated RPV prodrugs can extend the apparent drug half-life and improve tissue biodistribution. These results warrant further development for human use.
Keywords: Antiretroviral therapy; Long-acting slow effective release antiretroviral therapy; Monocyte-derived macrophages; Nanoformulation; Prodrugs; Rilpivirine.
Copyright © 2019 Elsevier B.V. All rights reserved.
Figures


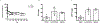
References
-
- May MT, Gompels M, Delpech V, Porter K, Orkin C, Kegg S, Hay P, Johnson M, Palfreeman A, Gilson R, Chadwick D, Martin F, Hill T, Walsh J, Post F, Fisher M, Ainsworth J, Jose S, Leen C, Nelson M, Anderson J, Sabin C, Study UKCHC, Impact on life expectancy of HIV-1 positive individuals of CD4+ cell count and viral load response to antiretroviral therapy, Aids, 28 (2014) 1193–1202. - PMC - PubMed
-
- Osterberg L, Blaschke T, Adherence to medication, The New England journal of medicine, 353 (2005) 487–497. - PubMed
-
- Winner B, Peipert JF, Zhao Q, Buckel C, Madden T, Allsworth JE, Secura GM, Effectiveness of long-acting reversible contraception, The New England journal of medicine, 366 (2012) 1998–2007. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
- R01 NS036126/NS/NINDS NIH HHS/United States
- R01 AI145542/AI/NIAID NIH HHS/United States
- P01 DA028555/DA/NIDA NIH HHS/United States
- P30 MH062261/MH/NIMH NIH HHS/United States
- T32 NS105594/NS/NINDS NIH HHS/United States
- R56 AI138613/AI/NIAID NIH HHS/United States
- P01 MH064570/MH/NIMH NIH HHS/United States
- R01 AG043540/AG/NIA NIH HHS/United States
- R01 NS034239/NS/NINDS NIH HHS/United States
- R01 MH115860/MH/NIMH NIH HHS/United States
- P01 NS043985/NS/NINDS NIH HHS/United States
- R01 AI158160/AI/NIAID NIH HHS/United States
- P01 NS031492/NS/NINDS NIH HHS/United States
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

