Titania nanotube-based protein delivery system to inhibit cranial bone regeneration in Crouzon model of craniosynostosis
- PMID: 31496688
- PMCID: PMC6690047
- DOI: 10.2147/IJN.S202090
Titania nanotube-based protein delivery system to inhibit cranial bone regeneration in Crouzon model of craniosynostosis
Abstract
Background: Craniosynostosis is a developmental disorder characterized by the premature fusion of skull sutures, necessitating repetitive, high-risk neurosurgical interventions throughout infancy. This study used protein-releasing Titania nanotubular implant (TNT/Ti) loaded with glypican 3 (GPC3) in the cranial critical-sized defects (CSDs) in Crouzon murine model (Fgfr2c342y/+ knock-in mutation) to address a key challenge of delaying post-operative bone regeneration in craniosynostosis.
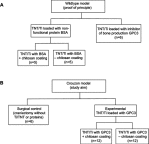
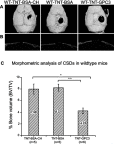
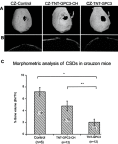
Materials and methods: A 3 mm wide circular CSD was created in two murine models of Crouzon syndrome: (i) surgical control (CSDs without TNT/Ti or any protein, n=6) and (ii) experimental groups with TNT/Ti loaded with GPC3, further subdivided into the presence or absence of chitosan coating (on nanotubes) (n=12 in each group). The bone volume percentage in CSDs was assessed 90 days post-implantation using micro-computed tomography (micro-CT) and histological analysis.
Results: Nano-implants retrieved after 90 days post-operatively depicted well-adhered, hexagonally arranged, and densely packed nanotubes with average diameter of 120±10 nm. The nanotubular architecture was generally well-preserved. Compared with the control bone volume percentage data (without GPC3), GPC3-loaded TNT/Ti without chitosan coating displayed a significantly lower volume percent in cranial CSDs (P<0.001). Histological assessment showed relatively less bone regeneration (healing) in GPC3-loaded CSDs than control CSDs.
Conclusion: The finding of inhibition of cranial bone regeneration by GPC3-loaded TNT/Ti in vivo is an important advance in the novel field of minimally-invasive craniosynostosis therapy and holds the prospect of altering the whole paradigm of treatment for affected children. Future animal studies on a larger sample are indicated to refine the dosage and duration of drug delivery across different ages and both sexes with the view to undertake human clinical trials.
Keywords: craniosynostosis; glypican; murine; protein delivery; titania nanotube.
Conflict of interest statement
The authors declare no conflicts of interest in this work.
Figures




Similar articles
-
Biological response of human suture mesenchymal cells to Titania nanotube-based implants for advanced craniosynostosis therapy.Colloids Surf B Biointerfaces. 2017 Feb 1;150:59-67. doi: 10.1016/j.colsurfb.2016.11.019. Epub 2016 Nov 17. Colloids Surf B Biointerfaces. 2017. PMID: 27883932
-
Glypican-based drug releasing titania implants to regulate BMP2 bioactivity as a potential approach for craniosynostosis therapy.Nanomedicine. 2018 Oct;14(7):2365-2374. doi: 10.1016/j.nano.2017.06.005. Epub 2017 Jun 23. Nanomedicine. 2018. PMID: 28648641
-
Cranial bone microarchitecture in a mouse model for syndromic craniosynostosis.J Anat. 2024 Dec;245(6):864-873. doi: 10.1111/joa.14121. Epub 2024 Aug 2. J Anat. 2024. PMID: 39096036 Free PMC article.
-
Titania nanotubes for orchestrating osteogenesis at the bone-implant interface.Nanomedicine (Lond). 2016 Jul;11(14):1847-64. doi: 10.2217/nnm-2016-0169. Epub 2016 Jul 7. Nanomedicine (Lond). 2016. PMID: 27389393 Review.
-
Local drug delivery to the bone by drug-releasing implants: perspectives of nano-engineered titania nanotube arrays.Ther Deliv. 2012 Jul;3(7):857-73. doi: 10.4155/tde.12.66. Ther Deliv. 2012. PMID: 22900467 Review.
Cited by
-
Targeting of C-ROS-1 Activity Using a Controlled Release Carrier to Treat Craniosynostosis in a Preclinical Model of Saethre-Chotzen Syndrome.J Tissue Eng Regen Med. 2024 May 9;2024:8863925. doi: 10.1155/2024/8863925. eCollection 2024. J Tissue Eng Regen Med. 2024. PMID: 40225751 Free PMC article.
-
Mechanical loading of cranial joints minimizes the craniofacial phenotype in Crouzon syndrome.Sci Rep. 2022 Jun 11;12(1):9693. doi: 10.1038/s41598-022-13807-9. Sci Rep. 2022. PMID: 35690633 Free PMC article.
-
Craniofacial therapy: advanced local therapies from nano-engineered titanium implants to treat craniofacial conditions.Int J Oral Sci. 2023 Mar 29;15(1):15. doi: 10.1038/s41368-023-00220-9. Int J Oral Sci. 2023. PMID: 36977679 Free PMC article. Review.
-
Erosion-inhibiting potential of the stannous fluoride-enriched CPP-ACP complex in vitro.Sci Rep. 2023 May 16;13(1):7940. doi: 10.1038/s41598-023-34884-4. Sci Rep. 2023. PMID: 37193788 Free PMC article.
References
-
- Gosain AK, McCarthy JG, Wisoff JH. Morbidity associated with increased intracranial pressure in Apert and Pfeiffer syndromes: the need for long-term evaluation. Plast Reconstr Surg. 1996;97(2):292–301. - PubMed
-
- Gonsalez S, Hayward R, Jones B, Lane R. Upper airway obstruction and raised intracranial pressure in children with craniosynostosis. Eur Respir J. 1997;10(2):367–375. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous

