Dual targeting curcumin loaded alendronate-hyaluronan- octadecanoic acid micelles for improving osteosarcoma therapy
- PMID: 31496695
- PMCID: PMC6691947
- DOI: 10.2147/IJN.S211981
Dual targeting curcumin loaded alendronate-hyaluronan- octadecanoic acid micelles for improving osteosarcoma therapy
Abstract
Introduction: Curcumin (CUR) is a general ingredient of traditional Chinese medicine, which has potential antitumor effects. However, its use clinically has been limited due to its low aqueous solubility and bioavailability. In order to improve the therapeutic effect of CUR on osteosarcoma (i.e., bone cancer), a multifunctional micelle was developed here by combining active bone accumulating ability with tumor CD44 targeting capacity.
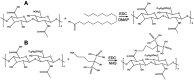
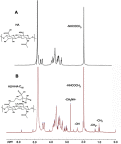
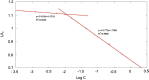
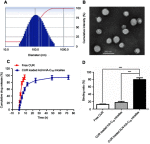
Methods: The CUR loaded micelles were self-assembled by using alendronate-hyaluronic acid-octadecanoic acid (ALN-HA-C18) as an amphiphilic material. The obtained micelles were characterized for size and drug loading. In addition, the in vitro release behavior of CUR was investigated under PBS (pH 5.7) medium containing 1% Tween 80 at 37℃. Furthermore, an hydroxyapatite (the major inorganic component of bone) affinity experiment was studied. In vitro antitumor activity was evaluated. Finally, the anti-tumor efficiency was studied.
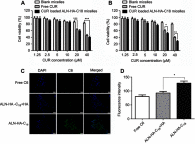
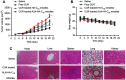
Results: The size and drug loading of the CUR loaded ALN-HA-C18 micelles were about 118 ± 3.6 nm and 6 ± 1.2%, respectively. CUR was released from the ALN-HA-C18 micelles in a sustained manner after 12 h. The hydroxyapatite affinity experiment indicated that CUR loaded ALN-HA-C18 micelles exhibited a high affinity to bone. CUR loaded ALN-HA-C18 micelles exhibited much higher cytotoxic activity against MG-63 cells compared to free CUR. Finally, CUR loaded ALN-HA-C18 micelles effectively delayed anti-tumor growth properties in osteosarcoma bearing mice as compared with free CUR.
Conclusion: The present study suggested that ALN-HA-C18 is a novel promising micelle for osteosarcoma targeting and delivery of the hydrophobic anticancer drug CUR.
Keywords: alendronate; curcumin; hyaluronic acid; osteosarcoma.
Conflict of interest statement
The authors report no conflicts of interest in this work.
Figures
Similar articles
-
Multifunctional redox-responsive and CD44 receptor targeting polymer-drug nanomedicine based curcumin and alendronate: synthesis, characterization and in vitro evaluation.Artif Cells Nanomed Biotechnol. 2018;46(sup1):168-177. doi: 10.1080/21691401.2017.1416390. Epub 2017 Dec 14. Artif Cells Nanomed Biotechnol. 2018. PMID: 29239219
-
In Vivo Evaluation of Reduction-Responsive Alendronate-Hyaluronan-Curcumin Polymer-Drug Conjugates for Targeted Therapy of Bone Metastatic Breast Cancer.Mol Pharm. 2018 Jul 2;15(7):2764-2769. doi: 10.1021/acs.molpharmaceut.8b00266. Epub 2018 Jun 1. Mol Pharm. 2018. PMID: 29792799
-
Alendronate-decorated biodegradable polymeric micelles for potential bone-targeted delivery of vancomycin.J Biomater Sci Polym Ed. 2015;26(11):629-43. doi: 10.1080/09205063.2015.1053170. Epub 2015 Jun 19. J Biomater Sci Polym Ed. 2015. PMID: 25994241
-
Prospects of Curcumin Nanoformulations in Cancer Management.Molecules. 2022 Jan 7;27(2):361. doi: 10.3390/molecules27020361. Molecules. 2022. PMID: 35056675 Free PMC article. Review.
-
Lipid-based nanodelivery systems of curcumin: Recent advances, approaches, and applications.Food Chem. 2025 Jan 15;463(Pt 2):141193. doi: 10.1016/j.foodchem.2024.141193. Epub 2024 Sep 11. Food Chem. 2025. PMID: 39276542 Review.
Cited by
-
Carboxymethyl Dextran-Based Nanomicelle Coatings on Microarc Oxidized Titanium Surface for Percutaneous Implants: Drug Release, Antibacterial Properties, and Biocompatibility.Biomed Res Int. 2022 Jul 12;2022:9225647. doi: 10.1155/2022/9225647. eCollection 2022. Biomed Res Int. 2022. PMID: 35865662 Free PMC article.
-
Harnessing curcumin and nanotechnology for enhanced treatment of breast cancer bone metastasis.Discov Nano. 2024 Nov 11;19(1):177. doi: 10.1186/s11671-024-04126-1. Discov Nano. 2024. PMID: 39527354 Free PMC article. Review.
-
CD44 In Sarcomas: A Comprehensive Review and Future Perspectives.Front Oncol. 2022 Jun 17;12:909450. doi: 10.3389/fonc.2022.909450. eCollection 2022. Front Oncol. 2022. PMID: 35785191 Free PMC article. Review.
-
Targeting bone in cancer therapy: Advances and challenges of bisphosphonate-based drug delivery systems.ADMET DMPK. 2025 Jun 8;13(3):2756. doi: 10.5599/admet.2756. eCollection 2025. ADMET DMPK. 2025. PMID: 40585412 Free PMC article. Review.
-
GO-Y078, a Curcumin Analog, Induces Both Apoptotic Pathways in Human Osteosarcoma Cells via Activation of JNK and p38 Signaling.Pharmaceuticals (Basel). 2021 May 24;14(6):497. doi: 10.3390/ph14060497. Pharmaceuticals (Basel). 2021. PMID: 34073773 Free PMC article.
References
-
- Nabavi SF, Daglia M, Moghaddam AH, Habtemariam S, Nabavi SM. Curcumin and liver disease: from chemistry to medicine. Compr Rev Food Sci Food Saf. 2013;13(1):62–77. - PubMed
-
- Aggarwal BB, Kumar ABharti AC. Anticancer potential of curcumin: preclinical and clinical studies. Anticancer Res. 2003;23(1A):363–398. - PubMed
-
- Lin JK, Lin-Shiau SY. Mechanisms of cancer chemoprevention by curcumin. Proc Natl Sci Counc Repub China B. 2001;25(2):59–66. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous

