The dynamic methylome of islets in health and disease
- PMID: 31500828
- PMCID: PMC6768570
- DOI: 10.1016/j.molmet.2019.06.007
The dynamic methylome of islets in health and disease
Abstract
Background: Epigenetic processes control timing and level of gene expression throughout life, during development, differentiation, and aging, and are the link to adapting gene expression profiles to environmental cues. To qualify for the definition of 'epigenetic', a change to a gene's activity must be inherited through at least one mitotic division. Epigenetic mechanisms link changes in the environment to adaptions of the genome that do not rely on changes in the DNA sequence. In the past two decades, multiple studies have aimed to identify epigenetic mechanisms, and to define their role in development, differentiation and disease.
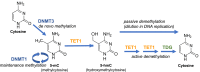
Scope of review: In this review, we will focus on the current knowledge of the epigenetic control of pancreatic beta cell maturation and dysfunction and its relationship to the development of islet cell failure in diabetes. Most of the data currently available have been obtained in mice, but we will summarize studies of human data as well. We will focus here on DNA methylation, as this is the most stable epigenetic mark, and least impacted by the variables inherent in islet procurement, isolation, and culture.
Major conclusions: DNA methylation patterns of beta cell are dynamic during maturation and during the diabetic process. In both cases, the changes occur at cell specific regulatory regions such as enhancers, where the methylation profile is cell type specific. Frequently, the differentially methylated regulatory elements are associated with key function genes such as PDX1, NKX6-1 and TCF7L2. During maturation, enhancers tend to become demethylated in association with increased activation of beta cell function genes and increased functionality, as indicated by glucose stimulated insulin secretion. Likewise, the changes to the DNA methylome that are present in pancreatic islets from diabetic donors are enriched in regulatory regions as well.
Keywords: Aging; Beta cells; DNA methylation; Endocrine pancreas; Epigenetics.
Copyright © 2019. Published by Elsevier GmbH.
Figures

References
-
- Waddington C.H. George Allen & Unwin Ltd; 1957. The strategy of genes.
-
- Talchai C., Lin H.V., Kitamura T., Accili D. Genetic and biochemical pathways of beta-cell failure in type 2 diabetes. Diabetes, Obesity and Metabolism. 2009;11:38–45. - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

