The fine-tuning of endoplasmic reticulum stress response and autophagy activation during trophoblast syncytialization
- PMID: 31501418
- PMCID: PMC6733854
- DOI: 10.1038/s41419-019-1905-6
The fine-tuning of endoplasmic reticulum stress response and autophagy activation during trophoblast syncytialization
Abstract
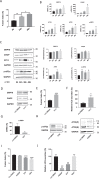
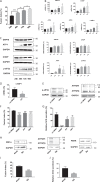
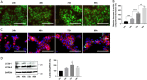
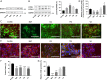
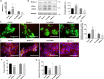
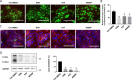
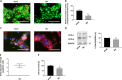
The syncytiotrophoblast (STB) is a multinuclear layer forming the outer surface of the fetal part of the placenta deriving from villous cytotrophoblastic cell (vCTB) fusion and differentiation. This syncytialization process is characterized by morphological and biochemical alterations of the trophoblast, which probably require removal of pre-existing structures and proteins to maintain cell homeostasis and survival. Interestingly, autophagy, which allows degradation and recycling of cellular components, was shown to be activated in syncytiotrophoblast. Here we examined the involvement of endoplasmic reticulum stress (ERS) response in autophagy activation during vCTB syncytialization. We first demonstrated the activation of ERS response and autophagy during the time course of trophoblastic cell fusion and differentiation. Alteration of autophagy activation in vCTB by chemical treatments or Beclin-1 expression modulation leads to a decrease in trophoblastic syncytialization. Furthermore, ERS response inhibition by chemical treatment or siRNA strategy leads to a default in syncytialization, associated with alteration of autophagy markers and cell survival. From these data, we suggest that ERS response, by fine regulation of autophagy activation, may serve as an adaptive mechanism to promote cell survival during trophoblastic syncytialization.
Conflict of interest statement
The authors declare that they have no conflict of interest.
Figures
References
-
- Baergen Rebecca N. Manual of Pathology of the Human Placenta. Boston, MA: Springer US; 2010. Chorionic Villi: Histology and Villous Development; pp. 69–83.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

