The frequency of circulating integrin α4β7+ cells correlates with protection against Helicobacter pylori infection in immunized mice
- PMID: 31502365
- PMCID: PMC6900148
- DOI: 10.1111/hel.12658
The frequency of circulating integrin α4β7+ cells correlates with protection against Helicobacter pylori infection in immunized mice
Abstract
Background: Chronic Helicobacter pylori infection is the cause of peptic ulcers in a subpopulation of individuals and a risk factor for the development of gastric cancer. A vaccine against H pylori infection can prevent the acquisition of the infection and protect against reinfections. Clinical trials to date evaluating the efficacy of H pylori vaccines in human challenge models have shown moderate to poor protection with difficulties in predicting efficacy. Thus, while further studies are needed to design an effective vaccine, we also need to find relevant correlates for vaccine efficacy.
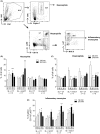
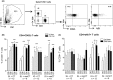
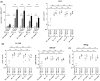
Objective: To find immune correlates to vaccine efficacy, the frequencies of neutrophils, eosinophils and inflammatory monocytes and CD4+ T-cell memory and mucosa homing integrin α4β7+ cells were assessed by flow cytometry in the blood of mice after vaccination.
Materials and methods: H pylori antigens and cholera toxin or the multiple mutant CT (mmCT) were administered via the sublingual (SL) and intragastric route (IG). The vaccinated mice were infected with H pylori strain SS1 bacteria, and colonization in the stomach and immune responses were evaluated.
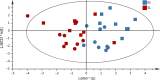
Results: The H pylori vaccine was effective in reducing bacterial load in the stomach of mice and enhancing immune responses compared to unvaccinated infection controls. In the blood of mice after SL or IG route of vaccination, we observed changes in frequencies of innate and adaptive immune cell subsets compared to infection controls. Remarkably, the frequency of circulating mucosal homing α4β7+ CD4+ T cells after vaccination correlated with low bacterial load in the stomach of individual mice irrespective of the immunization route.
Conclusions: Our study shows that the innate and adaptive immune cell subsets can be measured in the blood after vaccination and that increased frequency of α4β7+ CD4+ in the blood after immunization could be used as a predictive marker for the efficacy of vaccine against H pylori infection.
Keywords: Helicobacter pylori; CD4+T cells; IL-17A and IFNγ; inflammatory monocytes; neutrophils; α4β7.
© 2019 The Authors. Helicobacter published by John Wiley & Sons Ltd.
Figures

Similar articles
-
Induction of mucosal immune responses against Helicobacter pylori infection after sublingual and intragastric route of immunization.Immunology. 2017 Feb;150(2):172-183. doi: 10.1111/imm.12676. Epub 2016 Nov 7. Immunology. 2017. PMID: 27676456 Free PMC article.
-
Mucosal vaccination increases local chemokine production attracting immune cells to the stomach mucosa of Helicobacter pylori infected mice.Vaccine. 2012 Feb 21;30(9):1636-43. doi: 10.1016/j.vaccine.2011.12.111. Epub 2012 Jan 9. Vaccine. 2012. PMID: 22230589
-
Mucosal immunization with a urease B DNA vaccine induces innate and cellular immune responses against Helicobacter pylori.Helicobacter. 2006 Apr;11(2):113-22. doi: 10.1111/j.1523-5378.2006.00385.x. Helicobacter. 2006. PMID: 16579841
-
Inflammation, Immunity, and Vaccine Development for the Gastric Pathogen Helicobacter pylori.Curr Top Microbiol Immunol. 2019;421:1-19. doi: 10.1007/978-3-030-15138-6_1. Curr Top Microbiol Immunol. 2019. PMID: 31123883 Review.
-
Clearance of Helicobacter pylori infection through immunization: the site of T cell activation contributes to vaccine efficacy.Vaccine. 2004 Feb 17;22(7):888-97. doi: 10.1016/j.vaccine.2003.11.035. Vaccine. 2004. PMID: 15040942 Review.
Cited by
-
Targeting integrin pathways: mechanisms and advances in therapy.Signal Transduct Target Ther. 2023 Jan 2;8(1):1. doi: 10.1038/s41392-022-01259-6. Signal Transduct Target Ther. 2023. PMID: 36588107 Free PMC article. Review.
-
An Evaluation of Urease A Subunit Nanocapsules as a Vaccine in a Mouse Model of Helicobacter pylori Infection.Vaccines (Basel). 2023 Oct 27;11(11):1652. doi: 10.3390/vaccines11111652. Vaccines (Basel). 2023. PMID: 38005984 Free PMC article.
-
Developing a potent vaccine against Helicobacter pylori: critical considerations and challenges.Expert Rev Mol Med. 2024 Nov 25;27:e12. doi: 10.1017/erm.2024.19. Expert Rev Mol Med. 2024. PMID: 39584502 Free PMC article. Review.
References
-
- Ernst PB, Gold BD. The disease spectrum of Helicobacter pylori: the immunopathogenesis of gastroduodenal ulcer and gastric cancer. Annu Rev Microbiol. 2000;54:615‐640. - PubMed
-
- Ferlay J, Shin HR, Bray F, Forman D, Mathers C, Parkin DM. Cancer Incidence and Mortality Worldwide: IARC Cancer Base No. 10. GLOBCAN. 2008. Lyon: International Agency for Research on Cancer; 2010.
-
- Raghavan S, Holmgren J, Svennerholm A‐M. Chapter 51 – Helicobacter pylori infection of the gastric mucosa In: Strober W, Russell MW, Kelsall BL, Cheroutre H, Lambrecht BN, eds. Mucosal Immunology (4th ed.). Boston, MA: Academic Press; 2015:985‐1001.
-
- Czinn SJ, Blanchard T. Vaccinating against Helicobacter pylori infection. Nat Rev Gastroenterol Hepatol. 2011;8(3):133‐140. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials

