Myeloid loss of Beclin 1 promotes PD-L1hi precursor B cell lymphoma development
- PMID: 31503548
- PMCID: PMC6877338
- DOI: 10.1172/JCI127721
Myeloid loss of Beclin 1 promotes PD-L1hi precursor B cell lymphoma development
Abstract
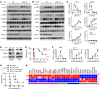
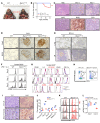
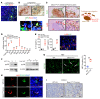
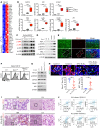
Beclin 1 (Becn1) is a key molecule in the autophagy pathway and has been implicated in cancer development. Due to the embryonic lethality of homozygous Becn1-deficient mice, the precise mechanisms and cell type-specific roles of Becn1 in regulating inflammation and cancer immunity remain elusive. Here, we report that myeloid-deficient Becn1 (Becn1ΔM) mice developed neutrophilia, were hypersusceptible to LPS-induced septic shock, and had a high risk of developing spontaneous precursor B cell (pre-B cell) lymphoma with elevated expression of immunosuppressive molecules programmed death ligand 1 (PD-L1) and IL-10. Becn1 deficiency resulted in the stabilization of MEKK3 and aberrant p38 activation in neutrophils, and mediated neutrophil-B cell interaction through Cxcl9/Cxcr3 chemotaxis. Neutrophil-B cell interplay further led to the activation of IL-21/STAT3/IRF1 and CD40L/ERK signaling and PD-L1 expression; therefore, it suppressed CD8+ T cell function. Ablation of p38 in Becn1ΔM mice prevented neutrophil inflammation and B cell tumorigenesis. Importantly, the low expression of Becn1 in human neutrophils was significantly correlated with the PD-L1 levels in pre-B acute lymphoblastic lymphoma (ALL) patients. Our findings have identified myeloid Becn1 as a key regulator of cancer immunity and therapeutic target for pre-B cell lymphomas.
Keywords: B cells; Cellular immune response; Immunology; Neutrophils; Oncology.
Conflict of interest statement
Figures



Comment in
-
Beclin-1 as a neutrophil-specific immune checkpoint.J Clin Invest. 2019 Dec 2;129(12):5079-5081. doi: 10.1172/JCI132534. J Clin Invest. 2019. PMID: 31657787 Free PMC article.
Similar articles
-
CXCL5 impedes CD8+ T cell immunity by upregulating PD-L1 expression in lung cancer via PXN/AKT signaling phosphorylation and neutrophil chemotaxis.J Exp Clin Cancer Res. 2024 Jul 22;43(1):202. doi: 10.1186/s13046-024-03122-8. J Exp Clin Cancer Res. 2024. PMID: 39034411 Free PMC article.
-
Beclin-1 as a neutrophil-specific immune checkpoint.J Clin Invest. 2019 Dec 2;129(12):5079-5081. doi: 10.1172/JCI132534. J Clin Invest. 2019. PMID: 31657787 Free PMC article.
-
Interferon-α Up-Regulates the Expression of PD-L1 Molecules on Immune Cells Through STAT3 and p38 Signaling.Front Immunol. 2018 Sep 27;9:2129. doi: 10.3389/fimmu.2018.02129. eCollection 2018. Front Immunol. 2018. PMID: 30356906 Free PMC article.
-
PD-1/PD-L1 Pathway and Its Blockade in Patients with Classic Hodgkin Lymphoma and Non-Hodgkin Large-Cell Lymphomas.Curr Hematol Malig Rep. 2020 Aug;15(4):372-381. doi: 10.1007/s11899-020-00589-y. Curr Hematol Malig Rep. 2020. PMID: 32394185 Review.
-
Prognostic and clinicopathological significance of PD-1/PD-L1 expression in the tumor microenvironment and neoplastic cells for lymphoma.Int Immunopharmacol. 2019 Dec;77:105999. doi: 10.1016/j.intimp.2019.105999. Epub 2019 Nov 6. Int Immunopharmacol. 2019. PMID: 31704289 Review.
Cited by
-
HDAC5 modulates PD-L1 expression and cancer immunity via p65 deacetylation in pancreatic cancer.Theranostics. 2022 Jan 31;12(5):2080-2094. doi: 10.7150/thno.69444. eCollection 2022. Theranostics. 2022. PMID: 35265200 Free PMC article.
-
Beclin1 controls caspase-4 inflammsome activation and pyroptosis in mouse myocardial reperfusion-induced microvascular injury.Cell Commun Signal. 2021 Nov 3;19(1):107. doi: 10.1186/s12964-021-00786-z. Cell Commun Signal. 2021. PMID: 34732218 Free PMC article.
-
Vangl2 suppresses NF-κB signaling and ameliorates sepsis by targeting p65 for NDP52-mediated autophagic degradation.Elife. 2024 Sep 13;12:RP87935. doi: 10.7554/eLife.87935. Elife. 2024. PMID: 39269442 Free PMC article.
-
The 'Danse Macabre'-Neutrophils the Interactive Partner Affecting Oral Cancer Outcomes.Front Immunol. 2022 Jun 16;13:894021. doi: 10.3389/fimmu.2022.894021. eCollection 2022. Front Immunol. 2022. PMID: 35784290 Free PMC article. Review.
-
RND2 attenuates apoptosis and autophagy in glioblastoma cells by targeting the p38 MAPK signalling pathway.J Exp Clin Cancer Res. 2020 Aug 31;39(1):174. doi: 10.1186/s13046-020-01671-2. J Exp Clin Cancer Res. 2020. PMID: 32867814 Free PMC article.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
Miscellaneous

