Programmed Chromosome Deletion in the Ciliate Oxytricha trifallax
- PMID: 31506317
- PMCID: PMC6778801
- DOI: 10.1534/g3.118.200930
Programmed Chromosome Deletion in the Ciliate Oxytricha trifallax
Abstract
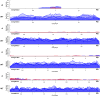
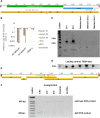
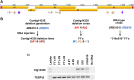
The ciliate Oxytricha trifallax contains two nuclei: a germline micronucleus and a somatic macronucleus. These two nuclei diverge significantly in genomic structure. The micronucleus contains approximately 100 chromosomes of megabase scale, while the macronucleus contains 16,000 gene-sized, high ploidy "nanochromosomes." During its sexual cycle, a copy of the zygotic germline micronucleus develops into a somatic macronucleus via DNA excision and rearrangement. The rearrangement process is guided by multiple RNA-based pathways that program the epigenetic inheritance of sequences in the parental macronucleus of the subsequent generation. Here, we show that the introduction of synthetic DNA molecules homologous to a complete native nanochromosome during the rearrangement process results in either loss or heavy copy number reduction of the targeted nanochromosome in the macronucleus of the subsequent generation. This phenomenon was tested on a variety of nanochromosomes with different micronuclear structures, with deletions resulting in all cases. Deletion of the targeted nanochromosome results in the loss of expression of the targeted genes, including gene knockout phenotypes that were phenocopied using alternative knockdown approaches. Further investigation of the chromosome deletion showed that, although the full length nanochromosome was lost, remnants of the targeted chromosome remain. We were also able to detect the presence of telomeres on these remnants. The chromosome deletions and remnants are epigenetically inherited when backcrossed to wild type strains, suggesting that an undiscovered mechanism programs DNA elimination and cytoplasmically transfers to both daughter cells during conjugation. Programmed deletion of targeted chromosomes provides a novel approach to investigate genome rearrangement and expands the available strategies for gene knockout in Oxytricha trifallax.
Keywords: Ciliates; Epigenetics; Genomic Rearrangement.
Copyright © 2019 Clay et al.
Figures



References
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
