G-CSF and GM-CSF Modify Neutrophil Functions at Concentrations found in Cystic Fibrosis
- PMID: 31506515
- PMCID: PMC6736848
- DOI: 10.1038/s41598-019-49419-z
G-CSF and GM-CSF Modify Neutrophil Functions at Concentrations found in Cystic Fibrosis
Abstract
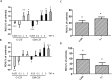
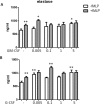
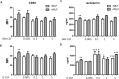
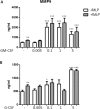
The role of colony stimulating factors (CSFs) in cystic fibrosis (CF) circulating neutrophils has not been thoroughly evaluated, considering that the neutrophil burden of lung inflammation in these subjects is very high. The aim of this study was to assess granulocyte-CSF (G-CSF) and granulocyte-macrophage-CSF (GM-CSF) levels in CF patients in various clinical conditions and how these cytokines impact on activation and priming of neutrophils. G-CSF and GM-CSF levels were measured in sputum and serum samples of stable CF patients (n = 21) and in CF patients with acute exacerbation before and after a course of antibiotic therapy (n = 19). CSFs were tested on non CF neutrophils to investigate their effects on reactive oxygen species (ROS) production, degranulation (CD66b, elastase, lactoferrin, MMP-9), and chemotaxis. At very low concentrations found in CF patients (0.005-0.1 ng/ml), both cytokines inhibited ROS production, while higher concentrations (1-5 ng/ml) exerted a stimulatory effect. While either CSF induced elastase and MMP-9 secretion, lactoferrin levels were increased only by G-CSF. Chemotaxis was inhibited by GM-CSF, but was increased by G-CSF. However, when present together at low concentrations, CSFs increased basal and fMLP-stimulated ROS production and chemotaxis. These results suggest the CSF levels that circulating neutrophils face before extravasating into the lungs of CF patients may enhance their function contributing to the airway damage.
Conflict of interest statement
The authors declare no competing interests.
Figures
Similar articles
-
Effects of granulocyte colony-stimulating factor and granulocyte-macrophage colony-stimulating factor on the bactericidal functions of neutrophils.Curr Opin Hematol. 1997 May;4(3):183-90. doi: 10.1097/00062752-199704030-00005. Curr Opin Hematol. 1997. PMID: 9209834 Review.
-
Recombinant human G-CSF and GM-CSF prime human neutrophils for superoxide production through different signal transduction mechanisms.J Lab Clin Med. 1991 Dec;118(6):576-84. J Lab Clin Med. 1991. PMID: 1720802
-
Cytotoxicity of white blood cells activated by granulocyte-colony-stimulating factor, granulocyte/macrophage-colony-stimulating factor and macrophage-colony-stimulating factor against tumor cells in the presence of various monoclonal antibodies.Cancer Immunol Immunother. 1994 Oct;39(4):254-62. doi: 10.1007/BF01525989. Cancer Immunol Immunother. 1994. PMID: 7525059 Free PMC article.
-
Effect of granulocyte and granulocyte macrophage colony stimulating factors (G-CSF and GM-CSF) on neonatal neutrophil functions.Pediatr Res. 2000 Sep;48(3):369-73. doi: 10.1203/00006450-200009000-00018. Pediatr Res. 2000. PMID: 10960505
-
Functional features of neutrophils induced by G-CSF and GM-CSF treatment: differential effects and clinical implications.Leukemia. 1997 Apr;11(4):466-78. doi: 10.1038/sj.leu.2400607. Leukemia. 1997. PMID: 9096685 Review.
Cited by
-
Neutrophils in COVID-19: Not Innocent Bystanders.Front Immunol. 2022 Jun 1;13:864387. doi: 10.3389/fimmu.2022.864387. eCollection 2022. Front Immunol. 2022. PMID: 35720378 Free PMC article. Review.
-
Multiparametric Profiling of Neutrophil Function via a High-Throughput Flow Cytometry-Based Assay.Cells. 2023 Feb 25;12(5):743. doi: 10.3390/cells12050743. Cells. 2023. PMID: 36899878 Free PMC article.
-
Hydroxychloroquine prophylaxis and treatment is ineffective in macaque and hamster SARS-CoV-2 disease models.JCI Insight. 2020 Dec 3;5(23):e143174. doi: 10.1172/jci.insight.143174. JCI Insight. 2020. PMID: 33090972 Free PMC article.
-
Airway epithelial cells mount an early response to mycobacterial infection.Front Cell Infect Microbiol. 2023 Sep 26;13:1253037. doi: 10.3389/fcimb.2023.1253037. eCollection 2023. Front Cell Infect Microbiol. 2023. PMID: 37822359 Free PMC article.
-
Immunogenic Properties of MVs Containing Structural Hantaviral Proteins: An Original Study.Pharmaceutics. 2022 Jan 1;14(1):93. doi: 10.3390/pharmaceutics14010093. Pharmaceutics. 2022. PMID: 35056989 Free PMC article.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Research Materials
Miscellaneous

